Conclusions
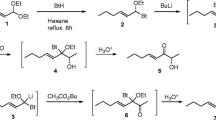
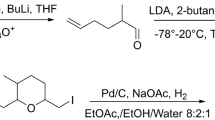
Simple routes for the synthesis of (2RS, 8R)-(−)-2-propionoxy-8-methyldecane (I) the pheromone of the leaf beetleDiabrotica virgifera virgifera — from chiral monofunctional synthons, obtained from the available (S)-(+)-3,7-dimethyl-1,6-octadiene, were developed.
Similar content being viewed by others
Literature cited
Nguen Kong Khao, M. V. Mavrov, and É. P. Serebryakov, Izv. Akad. Nauk SSSR, Ser. Khim., 1792 (1987).
K. Mori and H. Watanabe, Tetrahedron,40, 299 (1984).
Nguen Kong Khao, M. V. Mavrov, et al., Zh. Org. Khim., 1649 (1987).
Nguen Kong Khao, M. V. Mavrov, and É. P. Serebryakov, Izv. Akad. Nauk SSSR, Ser. Khim., 903 (1987).
G. Büchi and H. Wüester, J. Org. Chem.,34, 1122 (1969).
D. Thon and W. Schneider, Liebigs Ann. Chem., No. 11, 2094 (1976).
S. S. Poddubnaya, V. G. Cherkaev, and S. A. Voitkevich, Khimiya Drevesiny, No.4 93 (1983).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 9, pp. 2080–2083, September, 1987.
It corresponds to Communication 4 in the series “Terpenes in Organic Synthesis.” For the preceding Communication, cf. [1].
Rights and permissions
About this article
Cite this article
Khao, N.K., Mavrov, M.V. & Serebryakov, É.P. Coleopteran pheromones. Communication 9. Synthesis of the attractant of the leaf beetle diabrotica virgifera virgifera (coleoptera: Chrysamelidae) from (+)-3,7-di-methyl-1,6-octadiene. Russ Chem Bull 36, 1931–1934 (1987). https://doi.org/10.1007/BF00958349
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00958349