Abstract
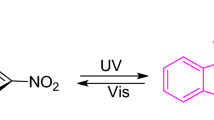
A study was carried out on a new class of photo- and thermochromic spirocyclic cyclohexadienoneperimidines and 4H-naphtholinoneperimidines. The structures of two of these compounds were determined. The lengths of the Cspiro-N bonds were found to be unequivalent; one of these bonds is cleaved in the photo- and thermochromic transformations. The nitrogen atom of the perimidine ring in both compounds was found to have pyramidal configuration with cis arrangement of the unshared electron pairs, while the cyclohexadienone fragments are strongly distorted toward the boat conformation. The nature of the dependence of the photo- and thermochromic properties on their structure and solvent was established.
Similar content being viewed by others
Literature cited
V. N. Komissarov, V. A. Kharlanov, L. Yu. Ukhin, and V. I. Minkin, Dokl. Akad. Nauk SSSR, 301, No. 4, 902 (1988).
V. N. Komissarov, V. A. Kharlanov, L. Yu. Ukhin, et al., Zh. Org. Khim., 26, No. 5, 1106–1110 (1990).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 3, pp. 702–708, March, 1991.
Rights and permissions
About this article
Cite this article
Aldoshin, S.M., Novozhilova, M.A., Atovmyan, L.O. et al. Synthesis, structure and properties of perimidine spirans. Russ Chem Bull 40, 617–623 (1991). https://doi.org/10.1007/BF00958007
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00958007