Abstract
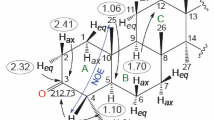
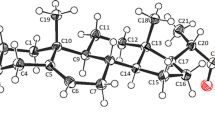
The validity of syn- and anti-isomer assignments in the case of 3Β,21-diacetoxypregna-5, 16-dien-20-one 20-carboethoxyhydrazones has been established by x-ray structural analysis; the isomer designations had been made previously based on chemical and spectral data. The isomers differ sharply in the conjugative relationship between the hydrazone group and the C16=C17 double bond in the steroid skeleton: the syn-isomer is trans-conjugated (the degree of twisting about the C17-C20 bond is characterized by a torsional angleψ=-161.0‡), while the anti-isomer is not conjugated (ϕ=− 75.3‡); this explains the electronic spectral features of these isomers.
Similar content being viewed by others
Literature cited
A. V. Kamernitskii, A. M. Turuta, Z. I. Istomina, and A. A. Korobov, Izv. Akad. Nauk SSSR, Ser. Khim., No. 1, 194 (1987).
V. S. Bogdanov, A. M. Turuta, and A. V. Kamernitskii, Izv. Akad. Nauk SSSR, Ser. Khim., No. 6, 1390 (1988).
A. V. Kamernitskii, N. S. Pavlova-Grishina, and A. V. Skorova, Izv. Akad. Nauk SSSR, Ser. Khim., No. 5, 1136 (1980).
Yu. P. Kitaev and B. I. Buzykin, Hydrazones [in Russian], Nauka, Moscow (1974), p. 48.
S. V. Lindeman, E. N. Tsapkina, Yu. T. Struchkov, et al., Bioorg. Khim.,14, No. 2, 261 (1988).
W. L. Duax and D. A. Norton, Atlas of Steroid Structure, Vol. 1, Plenum/IFI, New York (1975).
R. G. Gerr, A. I. Yanovskii, and Yu. T. Struchkov, Kristallografiya,28, No. 5, 1029 (1983).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 3, pp. 610–615, March, 1991.
Rights and permissions
About this article
Cite this article
Lindeman, S.V., Turuta, A.M., Struchkov, Y.T. et al. Molecular structures of syn- and anti-20-carboethoxyhydrazone isomers of 3Β, 21-diacetoxypregna-5,16-dien-20-one. Russ Chem Bull 40, 529–533 (1991). https://doi.org/10.1007/BF00957989
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957989