Abstract
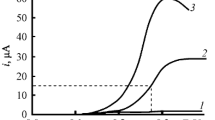
Oxidation of pyridine N-oxide in the presence of cyclohexane was studied on platinum and carbon glass rotating disk electrodes using the method of cyclic voltammetry in 0.2 N LiClO4 in acetonitrile. It was hypothesized that the electrochemically generated N-oxide radical cation reacts with cyclohexane according to the mechanism of hydrogen atom cleavage.
Similar content being viewed by others
Literature cited
Yu. V. Geletii, G. V. Lyubimova, and A. E. Shilov, Kinet. Katal.,26, No. 4, 1019 (1985).
S. I. Kulakovskaya, S. N. Shamaev, and V. M. Berdnikov, Izv. Akad. Nauk SSSR, Ser. Khim., No. 11, 2441 (1989).
Chemical Bond Rupture Energy. Ionization Potentials and Electron Affinity [in Russian], V. N. Kondrat'ev (ed.), Nauka, Moscow (1974).
S. I. Kulakovskaya, Yu. V. Geletii, L. A. Kushch, et al., Kinet. Katal.,28, No. 4, 1018 (1987).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 7, pp. 1488–1491, July, 1990.
Rights and permissions
About this article
Cite this article
Kulakovskaya, S.I., Shamaev, S.N. & Berdnikov, V.M. Mechanism of oxidation of cyclohexane by the radical cation of pyridine N-Oxide. Russ Chem Bull 39, 1345–1348 (1990). https://doi.org/10.1007/BF00957834
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957834