Abstract
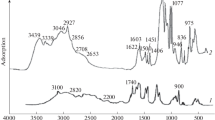
By means of kinetic studies and EPR, it has been shown that, in the reaction of hydrogen peroxide decomposition in the presence of copper-containing anion-exchange resins of the vinylpyridine series AN-40, AN-25, and AN-251, the catalytic activity increases in a series of complexes of the following types: Cu(Py)2 2+<Cu(Py)3 2+<Cu(Py)4 2+. Increases in the degree of polymer cross-linking and increases in the content of copper complexes above 0.2 mg-ion/g lead to the appearance of steric hindrance to free diffusion of H2O2 molecules within the globules of resin, with a consequent decrease in specific catalytic activity of the ion-exchanger/metal complexes. It has been established by means of EPR and IR spectroscopy that in AN-25 and AN-251 CH3 groups of the coordinated units are oxidized to carboxyl, with a consequent change in composition and structure of the Cu(II) complexes in the resins. Moreover, the copper ions are partly washed out from the resin into the reaction medium.
Similar content being viewed by others
Literature cited
V. D. Kopylova, E. L. Frumkina, L. A. Mochalova, and K. M. Saldadze, Kinet. Katal.,19, No. 5, 1356 (1978).
V. D. Kopylova, E. L. Frumkina, L. A. Mochalova, and K. M. Saldadze, Kinet. Katal.,21, No. 5, 1354 (1980).
E. L. Frumkina, Candidate's Dissertation, Moscow State University, Moscow (1985).
A. I. Kokorin, A. N. Astranina, V. D. Kopylova, et al., Izv. Akad. Nauk SSSR, Ser. Khim., No. 7, 1491 (1989).
A. I. Kokorin, K. I. Zamaraeva, V. Ya. Kovner, et al., Eur. Polym. J.,11, 719 (1975).
Yu. I. Skurlatov, A. I. Kokorin, S. O. Travin, et al., Vysokomol. Soedin., Ser. A,24, No. 9, 1874 (1982).
V. D. Kopylova, V. B. Kargman, L. N. Suvorova, et al., Zh. Fiz. Khim.,48, No. 6, 1500 (1974).
V. D. Kopylova, A. P. Rudenko, E. L. Frumkina, et al., Zh. Fiz. Khim.,57, No. 9, 2325 (1983).
A. P. Purmal', Doctoral Disseration, Institute of Chemical Physics, Academy of Sciences of the USSR, Moscow (1970).
V. V. Berentsveig, O. E. Dotsenko, A. I. Kokorin, et al., Izv. Akad. Nauk SSSR, Ser. Khim., No. 10, 2211 (1982).
A. I. Kokorin, V. V. Berentsveig, V. D. Kopylova, and E. L. Frumkina, Kinet. Katal.,24, No. 1, 181 (1983).
V. D. Skobeleva, I. P. Kolenko, L. S. Molochnikov, et al., Zh. Prikl. Khim.,60, No. 9, 2160 (1987).
V. D. Skobeleva, S. Yu. Men'shikov, L. S. Molochnikov, et al., Izv. Akad. Nauk SSSR, Ser. Khim., No. 8, 1728 (1987).
V. D. Laskorin, L. A. Fedorova, N. G. Zhukova, and V. A. Goldobina, Zh. Prikl. Khim.,47, No. 6, 1248 (1974).
A. L. Shvarts, V. B. Kargman, G. I. Zalkind, et al., Zh. Fiz. Khim.,55, No. 9, 2369 (1981).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 9, pp. 1970–1975, September, 1989.
Rights and permissions
About this article
Cite this article
Kokorin, A.I., Frumkina, E.L., Kopylova, V.D. et al. Decomposition of H2O2 on copper-containing ion-exchange resins of the vinylpyridine series. Russ Chem Bull 38, 1809–1814 (1989). https://doi.org/10.1007/BF00957767
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957767