Conclusions
-
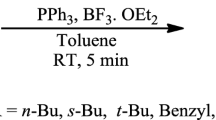
1.
Reaction of hexafluoropropylene oxide with DMF followed by treatment of the products with HCl gives perfluoro-4-oxo-2,5-dimethyl-2-fluorocarbonyl-1,3-dioxolane in high yield.
-
2.
Perfluoro-4-oxo-2,5-dimethyl-2-fluorocarbonyl-1,3-dioxolane reacts with fluoride ion at 20‡C with participation of only the fluorocarbonyl group forming cesium perfluoro-4-oxo-2,5-dimethyl-1,3-dioxolan-2-yl-methoxide. At higher temperatures ionic fragmentation of the ring proceeds with cleavage of the ester bond.
-
3.
Reaction of perfluoro-4-oxo-2,5-dimethyl-2-fluorocarbonyl-1,3-dioxolane with hexafluoropropylene oxide is a three-stage process with intermediate formation of the previously undescribed cesium perfluoro-A-oxo-2,5-dimethyl-1,3-dioxolan-3-yl-methoxide and perfluoro-4-oxo-2,5-dimethyl-2-(2′-oxa-3-fluorocarbonylbutyl)-1,3-dioxolane.
Similar content being viewed by others
Literature cited
Th. Croft and J. Brady, J. Fluor. Chem.,6, 213 (1975).
S. Selman, Patent 3,308,107, USA.
Th. Martini, Patent 2,623,089, West Germany.
I. L. Knunyants, N. I. Delyagina, and S. M. Igumnov, Izv. Akad. Nauk SSSR, Ser. Khim., 866 (1981).
D. Sianesi, A. Pasetti, and F. Tarli, Patent 1,503,361, France; Chem. Abstr.,70, P4100A.
S. Selmans, Patent 3,450,716, USA.
D. England, J. Fluor. Chem.,21, 377 (1982).
Th. Martini, VI Eur. Symp. Fluor. Chem. (1977).
T. G. Shibrya, S. P. Krukovskii, and V. A. Ponomarenko, Izv. Akad. Nauk SSSR, Ser. Khim., 1999 (1982).
L. Heinrich, Patent 81,856, East Germany.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 2, pp. 392–395, February, 1988.
Rights and permissions
About this article
Cite this article
Yuminov, V.S., Kartsov, S.V., Maksimov, V.L. et al. Perfluorinated dioxolanes 1. Synthesis of some derivatives of perfluoro-4-oxo-1,3-dioxolane. Russ Chem Bull 37, 311–315 (1988). https://doi.org/10.1007/BF00957432
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957432