Conclusions
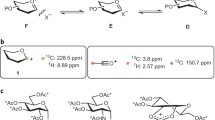
The behavior of acetates of glycofuranosyl fluorides under protolysis conditions in the gaseous phase is mainly determined by the relative configurations of the substituents at C1 and C4 atoms: the cis isomers form the [MH-HF]+ ions with higher probability, and the trans isomers, the [MH-AcOH]+ ions.
Similar content being viewed by others
Literature cited
V. I. Kadentsev, I. A. Trushkina, O. S. Chizhov, and Ya. V. Voznyi, Izv. Akad. Nauk SSSR, Ser. Khim., 2580 (1987).
Ya. V. Voznyi, I. S. Kalicheva, and A. A. Galoyan, Bioorg. Khim.,7, 406 (1981).
A. A. Solov'ev, V. I. Kadentsev, and O. S. Chizhov, Izv. Akad. Nauk SSSR, Ser. Khim., 2256 (1976).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 12, pp. 2708–2711, December, 1987.
Rights and permissions
About this article
Cite this article
Kadentsev, V.I., Trushkina, I.A., Chizhov, O.S. et al. Behavior of acetates of furanosyl fluorides under protolysis conditions in gaseous phase. Russ Chem Bull 36, 2510–2512 (1987). https://doi.org/10.1007/BF00957222
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957222