Conclusions
-
1.
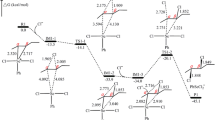
The MNDO method has been used to calculate sections of the potential energy surface for the dissociation of the anions CBr3 −, CHBr2, and CH2Br− to give the corresponding carbenes, and it is shown that the height of the activation barrier decreases with an increase in the number of Br atoms in the original carbanion and on passing to a more polar solvent.
-
2.
The presence of a counterion weakens the effect of solvent polarity on these reactions and impedes their occurrence in strongly polar media. A decrease in the radius of the counterion facilitates the occurrence of the reactions in solvents of weak polarity.
Similar content being viewed by others
Literature cited
I. A. Machkarovskaya, K. Ya. Burshtein, and V. A. Petrosyan, Izv. Akad. Nauk SSSR, Ser. Khim., No. 5, 1304 (1989).
M. J. S. Dewar and W. Thiel, J. Am. Chem. Soc.,99, 4899 (1977).
M. J. S. Dewar and E. Healy, J. Comput. Chem.,4, 542 (1983).
K. Ya. Burshtein, Zh. Strukt. Khim.,28, No. 2, 3 (1987).
K. Ya. Burshtein, J. Mol. Struct. Theochem.,153, 195, 203, 209 (1987).
M. Makosha and M. Fedoryn'ski, Zh. Vses. Khim. Ova.,24, No. 5, 466 (1979).
Author information
Authors and Affiliations
Additional information
For previous communication, see [1].
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 6, pp. 1309–1313, June, 1989.
Rights and permissions
About this article
Cite this article
Machkarovskaya, I.A., Burshtein, K.Y. & Petrosyan, V.A. Syntheses based on the electrogeneration of carbenes. 7. Kinetics of the formation of carbenes from the anions of bromomethanes. Russ Chem Bull 38, 1192–1196 (1989). https://doi.org/10.1007/BF00957152
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957152