Conclusions
-
1.
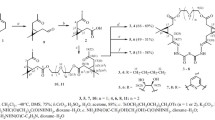
cis-Alloocimene adds acrylonitrile to give two stereoisomeric adducts, which exist in a half-chair conformation. Based on PMR spectra, one of the stereoisomers has a quasiaxial methyl group and pseudoequatorial nitrile and isopropylidene groups, while the other stereoisomer has a quasi-axial isopropylidene substituent and equatorial nitrile and methyl groups.
-
2.
Dichlorocyclopropanation of these adducts occurs from the sterically less hindered side of the molecules, with the formation, from one of the stereoisomers, of a cycloaddition product at the exocyclic double bond, and two stereoisomeric cycloaddition products at the two double bonds with a cis orientation of the cyclopropane ring and the CN group; the other stereoisomer, on the other hand, gives two stereoisomeric cycloaddition products at the two double bonds with a trans orientation of the cyclopropane ring and CN-group.
Similar content being viewed by others
Literature cited
B. A. Arbuzov and A. R. Vil'chinskaya, Zh. Obshch. Khim.,25, 168 (1955).
J. Chretien-Bessier, Ann. Chim., France, 301 (1957).
K. Alder, A. Dreike, and H. Erpenbach, Liebigs Ann. Chem.,609, 1 (1957).
A. R. Vil'chinskaya and B. A. Arbuzov, Zh. Obshch. Khim.,29, 2718 (1959).
J. E. Milks and J. E. Lankaster, J. Org. Chem.,30, 888 (1965).
E. K. V. Gustorf and I. Leitich, Tetrahedron Lett., 4689 (1968).
T. Sasaki, Sh. Equchi, H. Yamade, J. Org. Chem.,36, 1584 (1971).
C. Rucker, D. Lang, J. Sauer, et al., Chem. Ber.,113, 1663 (1980).
M. I. Karplus, Chem. Phys.,30, 11 (1959).
H. Booth, Tetrahedron,22, 615 (1966).
M. Anteunis, D. Tavernier, and F. Borremaus, Heterocycles,4, 293 (1976).
T. R. Forrest, J. Am. Chem. Soc.,97, 2628 (1975).
C. A. G. Haasnoot, F. A. A. M. Leeuw, H. P. M. Leeuw, and K. Altona, Rec. Trav. Chim. Pays-Bas.,98, 576 (1979).
I. B. Hendrickson, J. Am. Chem. Soc.,89, 7036 (1967).
V. A. Naumov, V. M. Bezzubov, N. M. Zaripov, and V. G. Dashevskii, Zh. Strukt. Khim.,11, 801 (1970).
G. Levy and G. Nelson, Handbook of Carbon-13 Nuclear Magnetic Resonance, Wiley, New York (1972).
F. Wehrli and I. Wirthlin, Interpretation of Carbon-13 NMR Spectra, Rheine-London-New York (1976).
B. A. Arbuzov, Zh. Obshch. Khim.,3, 21 (1933).
Author information
Authors and Affiliations
Additional information
Deceased.
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 5, pp. 1087–1093, May, 1988.
Rights and permissions
About this article
Cite this article
Arbuzov, B.A., Ratner, V.V., Danilova, O.I. et al. PMR study of the three-dimensional structure of stereoisomeric adducts of cis-alloocimene with acrylonitrile and their cyclization products with dichlorocarbene. Russ Chem Bull 37, 951–956 (1988). https://doi.org/10.1007/BF00957068
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957068