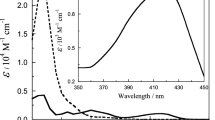
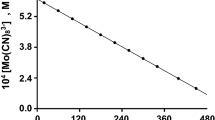
Conclusions
-
1.
Oxidation of the acetylacetonate ion in the coordination sphere of molybdenum(VI) by hydrogen peroxide proceeds without the participation of paramagnetic particles, recordable by steady-state ESR methods.
-
2.
It was established by the PMR method that the oxidation of 2,4-pentanedione by hydrogen peroxide in the presence of sodium molybdate proceeds through intermediate formation of the complex of Mo(VI) with 3,3-dihydroxy-2,4-pentanedione.
-
3.
A complex of Mo(VI) with 3,3-dihydroxy-2,4-pentanedione was produced for the first time.
Similar content being viewed by others
Literature cited
I. I. Moiseev, Zh. Vses. Khim. O-va.,22, 30 (1977).
O. Bortolini, F. Difia, Ch. Scardellato, and P. Sermin, J. Mol, Catal.,11, 107 (1981).
I. I. Moiseev, V. A. Bochkareva, A. E. Gekhman, Yu. V. Kokunov, and Yu. A. Buslaev, Dokl. Akad. Nauk SSSR,233, 375 (1977).
J. T. Spence, Coord. Chem. Rev.,48, 59 (1983).
M. Calvin and C. L. Wood, J. Am. Chem. Soc.,62, 3152 (1940).
A. J. Gordon and R. A. Ford, The Chemist's Companion: A Handbook of Practical Data, Techniques and References, Wiley (1972).
A. I. Busev, Analytical Chemistry of Molybdenum [in Russian], Izd, Akad. Nauk SSSR, Moscow (1962), p. 230.
G. Hesse and H. Stahl, Chem. Ber.,89, 2114 (1956).
B. M. Craven, K. G. Rameg, and W. B. Wise, Inorg. Chem.,10, 2626 (1971).
Yu. A. Buslaev, Yu. V. Kokunov, M. L. Gustyakova, Yu. D. Chubar, and I. I. Moiseev, Dokl. Akad. Nauk SSSR,233, 357 (1977).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 6, pp. 1242–1246, June, 1986.
The authors are grateful to E. P. Talzl for recording the ESR. spectra.
Rights and permissions
About this article
Cite this article
Gekhman, A.E., Nekipelov, V.M., Sakharov, S.G. et al. Mechanism of the oxidation of acetylacetone by hydrogen peroxide, catalyzed by Mo(VI) complexes. Russ Chem Bull 35, 1126–1130 (1986). https://doi.org/10.1007/BF00956580
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00956580