Conclusions
-
1.
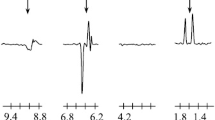
1H and13C NMR spectroscopy was used to study the photoconversion of methyl n-propyl ketone, methyl benzyl ketone, and methyl adamantyl ketone. The13C and1H CIDNP signs indicated that the polarization arises upon the cleavage of excited ketones in the triplet state.
-
2.
The competition of the radical photodecomposition (Norrish type-I) and “biradical” photodecomposition was studied in the photolysis of methyl n-propyl ketone. The ratio of the probabilities of the two radical pathways (recombination and disproportionation) is 3∶1, while the ratio of the two biradical pathways (photocyclization and Norrish type-II decomposition) is 3∶7.
-
3.
A mechanism was established for the steps in the conversions of the radicals and radical pairs arising in the photolysis of the ketones studied.
Similar content being viewed by others
Literature cited
G. Oster and N. L. Yang, Chem. Rev.,68, 125 (1968); S. Tazuke, Adv. Polym. Sci.,6, 321 (1969).
A. L. Buchachenko, É. M. Galimov, V. V. Ershov, G. A. Nikiforov, and A. D. Pershin, Dokl. Akad. Nauk SSSR,228, 379 (1976).
N. J. Turro, D. R. Anderson, and B. Kraeutler, Tetrahedron Lett.,21, 3 (1980).
J. Barltrop and J. Coyle, Excited States in Organic Chemistry, Wiley (1975).
A. L. Buchachenko, The Chemical Polarization of Electrons and Nuclei [in Russian], Nauka, Moscow (1974), p. 117.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 5, pp. 1024–1032, May, 1983.
Rights and permissions
About this article
Cite this article
Yankelevich, A.Z., Buchachenko, A.L., Potapov, V.K. et al. Chemically induced dynamic nuclear polarization and the mechanism for the photolysis of some dialkyl and aryl alkyl ketones. Russ Chem Bull 32, 927–934 (1983). https://doi.org/10.1007/BF00956141
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00956141