Conclusions
-
1.
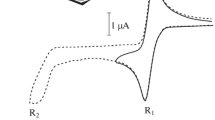
The complex metallocenyl chlorides Cp2MCl2 (M=Ti, Zr, Hf) are reversibly reduced in a THF medium on a dropping mercury electrode to the corresponding metastable radical anions\(Cp_2 MCl_2 \bar \cdot \), which are reversibly converted into the complexes Cp2MCl, the rate of the dehalo-genation reaction decreases as the temperature is lowered and increases along the series of metals Zr → Hf → Ti. The second reduction wave of the complexes Cp2MCl2 corresponds to the irreversible one-electron reduction of the complexes Cp2MCl2, which form in the layer next to the electrode as a result of the dehalogenation of\(Cp_2 MCl_2 \bar \cdot \), The third wave corresponds to the one-electron reduction of the radical anions\(Cp_2 MCl_2 \bar \cdot \) (M=Zr, Hf) or “titanocene” (M=Ti).
-
2.
The electroreduction of the complexes Cp2MCl2 to the M(III) and M(II) derivatives becomes increasingly hindered along the series Ti → Zr → Hf.
-
3.
The values of E1/2 of the complexes studied are linearly correlated with the fourth and third ionization potentials of the corresponding metals in the gaseous phase.
Similar content being viewed by others
Literature cited
N. M. Chirkov, P. E. Matkovskii, and F. S. D'yachkovskii, Polymerization on Complex Organometallic Catalysts [in Russian], Khimiya, Moscow (1976).
G. L. Soloveichik and B. M. Bulychev, Usp. Khim.,52, 72 (1983).
M. E. Vol'pin and V. B. Shur, “Fixation of molecular nitrogen in aprotic media,” in: New Trends in Chemical Nitrogen Fixation, Academic Press, London (1980), pp. 67–100.
E. Laviron, J. Besanson, and F. Hug, J. Organomet. Chem.,159, 279 (1978).
Y. Mugnier, C. Moise, and E. Laviron, J. Organomet. Chem.,204, 61 (1981).
Y. Mugnier, A. Fakhr, M. Fauconet, C. Moise, and E. Laviron, Acta Chim. Scand.,B37, 423 (1983).
N. El Murr, A. Chaloyard, and J. Tirouflet, J. Chem. Soc. Chem. Commun., 446 (1980).
V. V. Strelets, G. L. Soloveichik, A. I. Sizov, B. M. Bulychev, A. Rusina, and A. A. Vlchek, Izv. Akad. Nauk SSSR, Ser. Khim., 2493 (1983).
A. N. Nesmeyanov and K. A. Kochetkov (eds.), Methods of Organometallic Chemistry. The Subgroup of Copper, Scandium, Titanium, Vanadium, Chromium, and Manganese. The Lanthanides and Actinides [in Russian], Vol. 1, Nauka, Moscow (1974), pp. 181, 323.
V. V. Strelets, S. V. Kukharenko, G. L. Soloveichik, A. N. Protskii, and A. Rusina, Izv. Akad. Nauk SSSR, Ser. Khim., 1484 (1985).
A. Fakhr, Y. Mugnier, R. Broussier, B. Gautheron, and E. Laviron, J. Organomet. Chem.,255, C8 (1983).
H. P. Fritz, Adv. Organomet. Chem.,1, 240 (1964).
V. V. Strelets and S. V. Kukharenko, Dokl. Akad. Nauk SSSR,275, 894 (1984).
V. V. Strelets and S. V. Kukharenko, Nuov. J. Chim.,8, 785 (1984).
J. W. Lauher and R. Hoffmann, J. Am. Chem. Soc.,98, 1729 (1976).
S. Azavamuthan, C. Kalidas, and C. S. Venkatachalam, J. Electroanal. Chem.,171, 293 (1984).
S. Azavamuthan, C. Kalidas, and C. S. Venkatachalam, Ber. Bunsenges. Phys. Chem.,89, 880 (1985).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 5, pp. 1020–1027, May, 1986.
Rights and permissions
About this article
Cite this article
Kukharenko, S.V., Soloveichik, G.L. & Strelets, V.V. Electrochemistry of metallocene bichlorides of subgroup IVB in tetrahydrofuran. Russ Chem Bull 35, 926–932 (1986). https://doi.org/10.1007/BF00955351
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00955351