Conclusions
-
1.
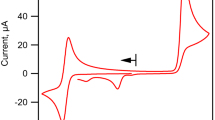
Electrolytic reduction of 3-formyl-[3]-ferrocenophane and acetylferrocene proceeds through the anion radical formation step.
-
2.
The influence of the structure on the behavior of the anion radicals being formed manifests itself only in nonaqueous solutions, in which the anion radical of acetylferrocene breaks down with rupture of the bond between the metal and the cyclopentadiene ring, whereas the anion radicals of [3]-ferrocenophanes retain the ferrocene structure, the break-down of which is prevented by the three-carbon bridge.
Similar content being viewed by others
Literature cited
V. P. Gul'tyai and A. M. Moiseenkov, Zh. Org. Khim.,16, 1026 (1980).
. M. Abbot, A. J. Bellamy, and J. Kerr, Chem. Ind., 828 (1974).
N. El Murr, A. Chaloyard, and E. Laviron, Nuovo Cimento,2, 15 (1975).
M. G. Peterleitner, L. I. Denisovich, A. Z. Kreindlin, and M. I. Rybinskaya, Izv. Akad. Nauk SSSR, Ser. Khim., No. 12, 780 (1981).
N. El Murr and A. J. Chaloyard, J. Organomet. Chem.,193, C60 (1980).
E. I. Denisovich and A. A. Koridze, Izv. Akad. Nauk SSSR, Ser. Khim., 276 (1981).
J. Heyrovsky and P. Zuman, Practical Polarography, Academic Press, New York (1968).
M. Lacon and Z. Ibrisagic, Croat. Chim. Acta,46, No. 2, 107 (1974).
A. J. Bellamy, J. B. Kerr, and I. S. Mackirdy, J. Chem. Soc. Perkin Trans. No. 2, 425 (1982).
R. G. Sutherland, J. R. Button, and W. M. Horspool, J. Organomet. Chem.,122, 393 (1976).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 12, pp. 2814–2818, December, 1983.
Rights and permissions
About this article
Cite this article
Peterleitner, M.G., Denisovich, L.I., Kreindlin, A.Z. et al. Comparative study of electrochemical reductions of 3-formyl-[3]-ferrocenophane and acetylferrocene. Russ Chem Bull 32, 2526–2529 (1983). https://doi.org/10.1007/BF00954487
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00954487