Conclusions
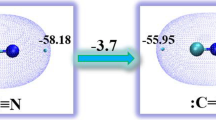
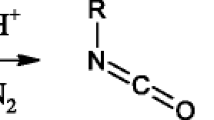
In addition to fragmentation at the C-N bond, XC6H4-CS-NH-C6H4Y thiobenzanilides form a set of fragments under electron impact which arise through a stage of isomerization of the molecular ion into a thiolimide, imidosulfide, and thiazirane structure.
Similar content being viewed by others
Literature cited
W. Walter and J. Voss, in: J. Zabicky (ed.), The Chemistry of Amides, Interscience Publishers, London-New York-Sydney-Toronto (1970), p. 383.
W. Walter, R. F. Becker, and H. F. Grützmacher, Tetrahedron Lett., 3513 (1968).
G. Harvath, Adv. Mass Spectrom.,6, 69 (1974).
T. J. Broxton and J. E. Rowe, Org. Mass Spectrom.,12, 185 (1977).
V. A. Puchkov, Yu. S. Nekrasov, and I. S. Vul'fson, Izv. Akad. Nauk SSSR, Ser. Khim., 1635 (1968).
L. A. Neiman, V. I. Maimind, M. M. Shemyakin, V. A. Puchkov, V. N. Bochkarev, Yu. S. Nekrasov, and N. S. Vul'fson, Zh. Obshch. Khim.,37, 1600 (1967).
R. H. Shapiro, J. W. Serum, and A. M. Duffield, J. Org. Chem.,33, 243 (1968).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 12, pp. 2740–2744, December, 1983.
Rights and permissions
About this article
Cite this article
Nekrasov, Y.S., Kandror, I.I., Vasyukova, N.I. et al. Behavior of thiobenzoic acid anilides under electron impact. Russ Chem Bull 32, 2457–2460 (1983). https://doi.org/10.1007/BF00954474
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00954474