Conclusions
-
1.
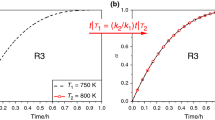
The kinetics, of quenching of the radicals formed under the effect of electron irradiation on liquid cyclohexanol and cyclododecanol in the absence of oxygen were investigated by pulsed radiolysis, and the absolute rate constants of α-hydroxycyclohexyl and α-hydroxycyclododecyl radicals in the 26–150°C and 85–135°C range were determined.
-
2.
The rates of quenching of these α-hydroxycycloalkyl radicals are limited by the rates of their chemical reaction.
Similar content being viewed by others
Literature cited
R. L. McCarthy and A. MacLachlan, Trans. Faraday Soc.,57, 1107 (1961).
N. Simic and H. Hayon, J. Phys. Chem.,75, 1677 (1971).
A. Shamin Chaudhri and K.-D. Asmus, J. Chem. Soc. Faraday Trans., 1,68, 385 (1972).
B. Ya. Ladygin, V. L. Mogilev, and M. S. Furman, Chemistry and Technology of Organic Synthesis Products [in Russian], 16th edn., Izd. GIAP, Moscow (1972), p. 31.
B. Ya. Ladygin, G. M. Zimina, and A. V. Vannikov, Izv. Akad. Nauk SSSR, Ser. Khim., 1018 (1984).
Handbook of Chemistry [in Russian], Khimiya, Leningrad (1971), p. 1072.
I. V. Khudyakov, P. P. Levin, and V. A. Kuz'min, Usp. Khim.,49, 1990 (1980).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 10, pp. 2194–2198, October, 1984.
Rights and permissions
About this article
Cite this article
Ladygin, B.Y., Zimina, G.M. & Revina, A.A. Kinetics of the reactions of radicals formed on pulsed electron radiation of cyclic alcohols in the liquid phase. Russ Chem Bull 33, 2001–2004 (1984). https://doi.org/10.1007/BF00954069
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00954069