Conclusions
-
1.
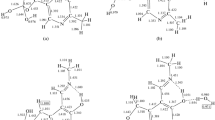
Quantum-chemical calculations have been used to establish the nature of the effect of the solvent on the enthalpy of reaction and the activation barrier in the dissociation of N-phosphorylammonium cations and zwitterions.
-
2.
It is shown that on passing from the gas phase to a polar solvent the strength of the phosphamide bond decreases in the cations, but increases in the zwitterions.
Similar content being viewed by others
Literature cited
V. F. Zarytova, Itogi Nauki. Tekh., Bioorg. Khim.,4, 102 (1984).
V. V. Pen'kovskii and I. E. Boldeskul, Teor. Éksp. Khim.,20, 198 (1984).
V. E. Klimenko, Teor. Éksp. Khim.,19, 612 (1983).
V. F. Zarytova, E. M. Ivanova, and V. P. Romanenko, Bioorg. Khim.,9, 516 (1983).
M. J. S. Dewar and W. Thiel, J. Am. Chem. Soc.,99, 4899 (1977).
A. Warshel, J. Phys. Chem.,83, 1640 (1979).
K. Ya. Burshtein, Zh. Strukt. Khim.,28, 3 (1987).
A. A. Bliznyuk and A. A. Voityuk, Zh. Strukt. Khim.,27, 190 (1986).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 2, pp. 315–318, February, 1989.
Rights and permissions
About this article
Cite this article
Mal'tseva, T.V., Voityuk, A.A. Quantum-chemical study of the effect of solvation on the strength of the phosphamide bond in N-phosphorylammonium cations and zwitterions. Russ Chem Bull 38, 267–270 (1989). https://doi.org/10.1007/BF00953611
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00953611