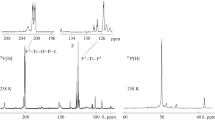
Conclusions
The relative stability of 1-substituted perfluoroallyl cations increases depending on the nature of the substituent in the following order: H < F < Cl < Br.
Similar content being viewed by others
Literature cited
M. V. Galakhov, V. A. Petrov, G. G. Belen'kii, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 1063 (1986).
M. V. Galakhov, G. G. Belen'kii, V. A. Petrov, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 1054 (1986).
M. H. Lien and A. C. Hopkinson, J. Phys. Chem.,88, 1513 (1984).
M. V. Galakhov, G. G. Belen'kii, V. A. Petrov, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 1057 (1986).
I. L. Knunyants, M. P. Krassuskaya, and E. I. Mysov, Izv. Akad. Nauk SSSR, Otd. Khim. Nauk, 1415 (1960).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 12, pp. 2761–2764, December, 1988.
Rights and permissions
About this article
Cite this article
Chepik, S.D., Galakhov, M.V., Belen'kii, G.G. et al. 13C- and19F-NMR spectra of 1-substituted analogs of the perfluoroallyl cation. Russ Chem Bull 37, 2486–2489 (1988). https://doi.org/10.1007/BF00952624
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00952624