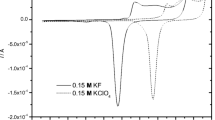
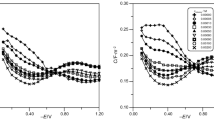
Conclusions
-
1.
By a special electrochemical method we have determined the parameters of monolayers of proteins adsorbed on a mercury surface: the surface concentration was 0.10–0.13 μg/cm2, the average thickness was 8–10 Å etc. — which agree with the parameters of irreversibly adsorbed proteins at boundaries of other phases. Proteins adsorbed on the mercury surface in the monolayer are found in the denatured, conformationally-flattened state.
-
2.
For low degrees of coverage and at the beginning of accumulation of protein on the mercury surface, deep conformational unfolding of the globular proteins occurs down to a thickness of the polypeptide chain, 5–6 Å.
-
3.
In the monolayer, the film of flattened protein is found in a compressed state; in this case, part of the segments of the polypeptide chain are displaced into the volume and formation of a secondary structure occurs. Compression and extension of the protein film is accompanied by fast and reversible rearrangement of the chain segments.
-
4.
Surface denaturation and irreversible adsorption of proteins are the principal reasons for the strong inhibition of electrochemical reactions of proteins.
Similar content being viewed by others
Literature cited
I. V. Berezin, V. A. Bogdanovskaya, S. D. Varfolomeev, M. R. Tarasevich, and A. I. Yaropolov, Dokl. Akad. Nauk SSSR,240, 615 (1978).
F. Scheller, R. Reuneberg, G. Strand, K. Pommerening, and P. Mohr, Bioelectrochem. Bioenerg.,4, 500 (1977).
S. D. Varfolomeev, in: Summaries of Science and Technology. Biological Chemistry [in Russian], Nauka, Moscow (1978), Vol. 12, p. 253.
B. A. Kuznetsov, Dokl. Akad. Nauk SSSR,195, 986 (1970).
B. A. Kuznetsov, N. M. Mestechkina, and G. P. Shumakovich, Bioelectrochem. Bioenerg.,4, 1 (1977).
G. P. Shumakovich and B. A. Kuznetsov, Biofizika,24, 777 (1979).
F. Schneller, M. Jänchen, and H. J. Prümke, Biopolymers,14, 1553 (1975).
S. R. Betso, M. H. Klapper, and L. B. Anderson, J. Am. Chem. Soc.,94, 8197 (1972).
B. A. Kuznetsov, G. P. Shumakovich, and N. M. Mestechkina, Bioelectrochem. Bioenerg.,4, 512 (1977).
R. Brdicka, Research,1, 25 (1947).
D. F. Cheesman and J. T. Davies, Advances Protein Chem.,9, 439 (1954).
L. K. James and L. G. Augenstein, Advances Enzymology,28, 1 (1966).
A. Rothen, Progr. Surface and Membr. Sci.,8, 81 (1974).
R. M. Azzam, J. Rigby, G. Perry, and J. A. Rueger, Phys. Med. Biol.,22, 422 (1977).
B. A. Kuznetsov and G. P. Shumakovich, Dokl. Akad. Nauk SSSR,210, 1223 (1973).
W. Morrissey and R. R. Stromberg, J. Colloid. Interface Sci.,46, 152 (1974).
M. C. Phillips, M. T. Evans, D. E. Graham, and D. Oldanio, Colloid. Polym. Sci.,253, 424 (1975).
F. R. Eirich, J. Colloid. Interface Sci.,58, 423 (1977).
E. Nyilas, T. H. Chin, and D. M. Lederman, in: Colloid. Interface Sci., Academic Press, New York (M. Kerker, ed.), Vol. 5 (1976), p. 77.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 10, pp. 2282–2290, October, 1982.
Rights and permissions
About this article
Cite this article
Kuznetsov, B.A., Shumakovich, G.P. Voltammetric study of conformational changes and properties of a protein monolayer adsorbed on a mercury electrode. Russ Chem Bull 31, 2009–2015 (1982). https://doi.org/10.1007/BF00950643
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00950643