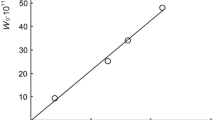
Conclusions
-
1.
The character of chain termination in the oxidation ofβ-ionone changes from quadratic to linear as the temperature increases, and the reaction rate has a negative temperature coefficient in the transitional region.
-
2.
The kinetic parameters of the oxidation rate ofβ-ionone have been determined.
Similar content being viewed by others
Literature cited
O. T. Kasaikina, A. B. Gagarina, and N. M. Émanuél', Izv. Akad. Nauk SSSR, Ser. Khim. 2243 (1975).
N. M. Evteeva, A. B. Gagarina, and N. M. Émanuél, Dokl. Akad. Nauk SSSR,224, 365 (1975);249, 637 (1979).
J. P. Van Hook and A. V. Tobolsky, J. Am. Chem. Soc.,80, 779 (1958).
Kh. S. Bagdasar'yan and R. I. Milyutinskaya, Zh. Fiz. Khim.,27, 420 (1953).
P. A. Ivanchenko, V. V. Kharitonov, and E. T. Denisov, Vysokomol. Soedin.,A11, 1622 (1969).
V. L. Rubailo, A. B. Gagarina, and N. M. Émanuél, Dokl, Akad. Nauk SSSR,213, 151 (1973);224, 4 (1975).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 12, pp. 2715–2720, December, 1980.
Rights and permissions
About this article
Cite this article
Evteeva, N.M., Gagarina, A.B. Temperature-dependent change in the chain-termination mechanism in the oxidation of β-ionone. Russ Chem Bull 29, 1891–1895 (1980). https://doi.org/10.1007/BF00949651
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00949651