Conclusions
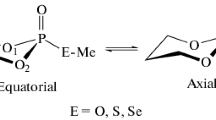
In the equilibrium of 5-phenyl-2,4,6-triisopropyl-1,3,5-dioxaphosphorinane and its derivatives the stereoisomer with nonequivalent protons on the carbon atoms attached to phosphorus is found predominantly in the twist form.
Similar content being viewed by others
Literature cited
B. A. Arbuzov, O. A. Erastov, and S. Sh. Khetagurova, Dokl. Akad. Nauk SSSR,246, 326 (1979).
B. A. Arbuzov, O. A. Erastov, S. Sh. Khetagurova, and T. A. Zyablikova, Dokl. Akad. Nauk SSSR,244, 610 (1979).
Yu. Yu. Samitov, Atlas of NMR Spectra of Steric Isomers [in Russian], Izd. Kazansk. Univ. (1978), p. 9.
B. A. Arbuzov, O. A. Erastov, S. Sh. Khetagurova, R. P. Arshinova, R. A. Kadyrov, and V. N. Smirnov, Izv. Akad. Nauk SSSR, Ser. Khim.,1979, 2239.
A. B. Pepperman, Jr., G. J. Boudreaux, and T. H. Siddall, J. Org. Chem.,40, 2056 (1975).
A. N. Vereshchagin and L. A. Grozina, Teor. Eksp. Khim.,4, 361 (1968).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 7, pp. 1630–1634, July, 1980.
Rights and permissions
About this article
Cite this article
Arbuzov, B.A., Erastov, O.A., Khetagurova, S.S. et al. Twist form in equilibrium of stereoisomers of 5-phenyl-2,4,6-triisopropyl-1,3,5-dioxaphosphorinane and its derivatives. Russ Chem Bull 29, 1174–1177 (1980). https://doi.org/10.1007/BF00949178
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00949178