Conclusions
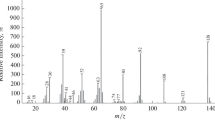
The method of electron impact has been used to obtain new data and to refine the known data on the processes of ion formation in the mass spectrum of oxygen difluoride. It has been confirmed that the thermal decomposition of oxygen difluoride includes a step with the formation of oxygen monofluoride. The formation of HgOF in the vapor has been postulated. A value of EA(FO) has been recommended for practical use.
Similar content being viewed by others
Literature cited
V. H. Dibeler, R. M. Reese, and J. L. Franklin, J. Chem. Phys.,27, 1296 (1957).
A. B. Cornford, D. C. Frost, F. G. Herring, and C. A. McDowell, J. Chem. Phys.,55, 2820 (1971).
J. Berkowitz, P. M. Dermer, and W. A. Chupka, J. Chem. Phys.,59, 925 (1973).
V. I. Alekseev, L. I. Fedorova, and A. V. Baluev, Izv. Akad. Nauk SSSR, Ser. Khim., 1084 (1983).
V. N. Kondrat'ev (ed.). Handbook of Bond Energies, lonization Potentials, and Electron Affinity [in Russian], Nauka, Moscow (1974).
W. M. Hickam and R. E. Fox, J. Chem. Phys.,25, 642 (1956).
J. M. Dyke, N. Jonathan, J. D. Mills, and A. Morris, Mol. Phys.,40, 1177 (1980).
V. P. Glushko, L. V. Gurvich, et al., Thermodynamic Properties of Individual Substances [in Russian], Book 2, Nauka, Moscow (1978).
J. Czarnowski and H. J. Schumacher, Chem. Phys. Lett.,17, 235 (1972).
C. E. Berry, Phys. Rev.,78, 597 (1950).
P. W. Harland and J. L. Franklin, J. Chem. Phys.,61, 1621 (1973).
P. A. G. O'Hary and A. C. Wahl, J. Chem. Phys.,53, 2469 (1970).
A. C. Wahl, P. Bertoncini, K. Kaiser, and R. Land, J. Quant. Chem.,35, 499 (1970).
A. S. Zyubin and O. P. Charkin, Zh. Strukt. Khim.,21, 3 (1980).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 6, pp. 1302–1306, June, 1984.
We thank Prof. L. N. Gorokhov for participating in the discussion of the results.
Rights and permissions
About this article
Cite this article
Alekseev, V.I., Volkov, V.M., Fedorova, L.I. et al. Mass-spectrometric investigation of the ionization processes of the oxygen difluoride molecule. Russ Chem Bull 33, 1195–1199 (1984). https://doi.org/10.1007/BF00948986
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00948986