Conclusions
-
1.
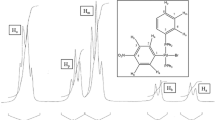
When oxidants act on palladium(II) chelates with 1,2-hydroxylaminooximes, oxidative hydrogenation occurs via the N=H bond of the coordinated ligands.
-
2.
It has been shown by EPR that an intermediate in the oxidation is a palladium(II) complex that contains a nitroxyl radical.
Similar content being viewed by others
Literature cited
S. A. D'yachenko, A. I. Stetsenko, L. B. Volodarskii, G. P. Zlobina, L. P. Sharova, and M. V. Shirai, Koordinats. Khim.,6, 1051 (1980).
S. V. Larionov, V. N. Kirichenko, A. I. Stetsenko, K. P. Naumova, S. A. D'yachenko, and L. B. Volodarskii, Zh. Neorg. Khim.,28, 411 (1983).
K. P. Naumova, A. I. Stetsenko, and L. B. Volodarskii, Zh. Obshch. Khim.,49, 2564 (1979).
Dictionary of Organic Compounds, Vol. 3 [Russian translation], Inostr. Lit., Moscow (1949), p. 684.
A. G. Milaev and O. Yu. Okhlobystin, Usp. Khim.,49, 1829 (1980).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 11, pp. 2592–2596, November, 1984.
Rights and permissions
About this article
Cite this article
Larionov, S.V., Kosareva, L.A. & Boguslavskii, E.G. Oxidation of palladium(II) chelates with 1,2-hydroxylaminooximes. Russ Chem Bull 33, 2374–2377 (1984). https://doi.org/10.1007/BF00948856
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00948856