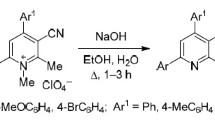
Abstract
The dihydropyridazine ring undergoes contraction to a dihydropyrrole ring to give 1-phenyl-2-benzylideneaminodihydropyrrolo[3,4-b]indol-3-ones when 1-phenyldihydropyridazino[4,5-b]indol-4-ones are treated with aromatic aldehydes under acid catalysis conditions. The reaction mechanism consists in the formation of a (3-indolyl)phenylmethyl cation, which leads to opening of the pyridazine ring and subsequent development of a bond between the carbonium center and the amide nitrogen atom.
Similar content being viewed by others
Literature cited
A. Alemany, M. Bernabe, C. Elorriaga, E. F. Alvarez, M. Lora-Tamaya, and O. Nieto, Bull. Soc. Chim. France,8, 2486 (1966).
R. Elderfield (editor), Heterocyclic Compounds, Vol. 8, Wiley (1950–1967).
N. A. Kogan, Reakts. Sposobn. Org. Soedin.,11, No. 3, 659 (1975).
M. I. Vlasova and N. A. Kogan, Khim. Geterotsikl. Soedin., No. 9, 1218 (1976).
N. A. Kogan and M. I. Vlasova, Khim. Geterotsikl. Soedin., No. 12, 1654 (1973).
N. A. Kogan and M. I. Vlasova, Khim.-Farmats. Zh., No. 7, 27 (1971).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1406–1408, October, 1976.
Rights and permissions
About this article
Cite this article
Kogan, N.A., Vlasova, M.I. Isomerization of pyridazino [4,5-b] indoles to pyrrolo [3,4-b] indoles. Chem Heterocycl Compd 12, 1162–1165 (1976). https://doi.org/10.1007/BF00945606
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00945606