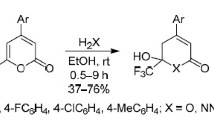
Abstract
A method has been developed for obtaining indolo-α-pyrones by acylation of 3-indolylacetic acid and its N-methyl derivatives. Indolo-α-pyrones form indolo-α-pyridones by refluxing with ammonium acetate in acetic acid. Indolo[2,3-c]pyrylium salts were obtained by acylation of 4-methoxyand 3,4-dimethoxy-3-phenacylindoles.
Similar content being viewed by others
Literature cited
S. Sakurai and T. Ito, Nippon Kagaku Zasshi,78, 1665 (1957); C., 14,046 (1958).
H. Pleninger, W. Muller, and K. Weinert, Chem. Ber.,95, 667 (1964).
G. N. Dorofeenko and V. G. Korobkova, Chem. Ind., 1848 (1968).
A. Baeyer, Ber.,42, 2337 (1910).
R. H. Wiley and I. C. Esterle, J. Org. Chem.,22, 1257 (1957).
V. Prey, B. Kerres, and H. Berbal, Monatsh.,91, 774 (1960).
H. E. Baumgarten, W. F. Murdock, and I. E. Dirks, J. Org. Chem.,26, 803 (1961).
G. N. Dorofeenko, L. V. Dulenko, V. I. Dulenko, and S. V. Krivun, Zh. Organ. Khim.,1, 117 (1965).
G. N. Dorofeenko and L. V. Dulenko, Khim. Geterotsikl. Soedin., 417 (1969).
A. T. Balaban, G. D. Mateescu, and M. Elian, Tetrahedron, 1083 (1962).
A. D. Semenov, G. N. Dorofeenko, and V. I. Dulenko, Khim. Geterotsikl. Soedin., 14 (1966).
R. C. Gilmore and W. I. Horton, J. Am. Chem. Soc.,73, 1411 (1951).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 345–349, March, 1971.
Rights and permissions
About this article
Cite this article
Dorofeenko, G.N., Korobkova, V.G. & Guzhina, E.A. Synthesis of indolo-α-pyrones, indolo-α-pyridones, and indolopyrylium salts. Chem Heterocycl Compd 7, 319–323 (1971). https://doi.org/10.1007/BF00944412
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00944412