Abstract
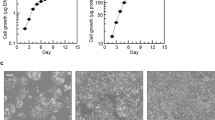
N-6 fatty acid metabolism was compared in NIH-3T3 cells and DT cells, which differ only in the presence of the v-Ki-ras oncogene. Non-dividing cells were incubated with [1-14C]-labelled fatty acids (18:2n-6, 18:3n-6, 20:3n-6 and 20:4n-6) at different time intervals (2–24 h) and concentration (0–120 μM). In both cells lines, the uptake of different fatty acids from the medium was similar and reached a maximum at 6–8 h. All fatty acids reached the same maximum level in DT cells, whereas, the relative uptake of added fatty acids by NIH-3T3 cells was different: 20:4n-6>20:2n-6>18:2n-6=18:3n-6. Throughout the incubation (2–24 h), desaturation and elongation of n-6 fatty acids was more active in DT cells than in NIH-3T3 cells. However, in both cell lines, incubated with different n-6 fatty acid precursors, the levels of radiolabelled 20:4n-6 were relatively constant. In DT cells, phosphatidylcholine was found to be the major fraction labelled with n-6 fatty acids precursors and those of endogenous synthesis, whereas, in NIH-3T3 cells the neutral lipid fraction, particularly triglycerides, was also strongly labelled. In concentration dependent studies, phospholipid labelling by fatty acids was saturable. At lower concentrations, especially in DT cells, phospholipids were labelled predominantly. As the concentration increased there was an overflow into the triglyceride fraction. Since the differences in fatty acid metabolism between the two cell lines cannot be related to the growth rate, it is suggested that they were a consequence of the expression of the v-Ki-ras oncogene.
Similar content being viewed by others
Abbreviations
- BSA:
-
bovine serum albumin
- CE:
-
cholesterol ester
- DG:
-
diglyceride
- DMEM:
-
Dulbecco's modification of Eagle's medium
- EL:
-
ether lipids (glyceryl ether diesters)
- FAME:
-
fatty acid methyl ester
- FCS:
-
fetal calf serum
- FFA:
-
free fatty acids
- HEPES:
-
N-2-(hydroxyethyl)piperazine-N′-2-ethanesulphonic acid
- MG:
-
monoglyceride
- NL:
-
neutral lipid
- PC:
-
phosphatidylcholine
- PE:
-
phosphatidylethanolamine
- PI:
-
phosphatidylinositol
- PL:
-
phospholipid
- s.a:
-
specific activity
- TG:
-
triglyceride
- TLC:
-
thin layer chromatography
References
Punnonen K, Puustin T, Koulu L, Jansen CT: Incorporation and distribution of eicosanoid precursor fatty acids in murine fibroblast in culture. Acta Physiol Scand 131: 583–587, 1987
Alonso T, Santos E: Increased intracellular glycerophosphoinositol is biochemical marker for transformation by membrane-associated and cytoplasmic oncogenes. Biochem Biophys Res Commun 171: 14–19, 1990
Benjamin CW, Gary Tapley W, Corman RR: The lack of PDGF-stimulated PGE2 release from ras-transformed NIH-3T3 cells results from reduced phospholipase C but not phospholipase A2 activity. Biochem Biophys Res Commun 145: 1254–1259, 1987
Noda M, Selinger Z, Scolnick EM, Bassin RH: Flat revertants isolated from Kirsten sarcoma virus-transformed cells are resistant to the action of specific oncogenes. Proc Natl Acad Sci USA 80: 5602–5606, 1983
Toth S, Fu T, Nozawa Y, Csaba G: The effect of epinephrine on the intracellular free calcium of parent and Ki-ras-transfected NIH3T3 cells. Biochem Biophys Res Commun 190: 353–361, 1993
Teegarden D, Taparowsky EJ, Kent C: Altered phosphatidylcholine metabolism in C3H10T1/2 cells transfected with the Harvey-ras oncogene, J Biol Chem 265: 6042–6047, 1990
Wakelam MJO, Davies SA, Houslay MD, McKay I, Marshall CJ, Hall A: Normal p21N-ras couples bombesin and other growth factor receptors to inositol phosphate production. Nature 323: 173–176, 1986
Cantrill RC, Ells GW, deAntueno RJ, Elliot M, Raha SK, Horrobin DF: The effect of n-6 fatty acids on normal and v-Ki ras transformed NIH-3T3 cells. Anticancer Res 12: 2197–2202, 1992
Cook HW, Clarke JTR, Spence MW: Concerted stimulation and inhibition of desaturation, chain elongation, and esterification of essential fatry acids by cultured neuroblastoma cells. J Biol Chem 258: 7586–7591, 1983
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ: Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275, 1951
Stewart JCM: Colorimetric determination of phospholipids with ammonium ferrothiocyanate. Anal Chem 104: 10–14, 1980
Folch J, Lees M, Sloane-Stanley GA: A simple method for the isolation and purfication of total lipides from animal tissues. J Biol Chem 226: 497–509, 1957
Snyder F, Wood R: The occurence and metabolism of alkyl and alk-1-enyl ethers of glycerol in transplantable rat mouse tumors. Cancer Res 28: 972–978, 1968
Chakravarthy BR, Spence MW, Clarke JTR, Cook HW: Rapid isolation of neuroblastoma plasma membranes on Percoll gradients. Characterization and lipid composition. Biochim Biophys Acta 812: 223–233, 1985
Morrison WR, Smith LM: Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride-methanol. J Lipid Res 5: 600–608, 1964
de Antueno RJ, Cantrill RC, Huang YS, Elliot M, Horrobin DF: Relationship between mouse liver Δ9 desaturase activity and plasma lipids. Lipids 28: 285–290, 1993
Gaspar G, de Alaniz MJT, Brenner R: Incorporation and metabolism of stearic, oleic, linoleic and α-linolenic acids in minimal deviation Hepatoma 7288C cells. Mol Cell Biochem 16: 197–203, 1977
Gaspar G, de Alaniz MJT, Brenner R: Uptake and metabolism of exogenous eicosa 8, 11, 14-trienoic acid in minimal deviation Hepatoma 7288 C cells. Lipids 10: 726–731, 1975
Chapkin RS, Hubbard NE, Buckman EK, Erickson KL: Linoleic acid metabolism in metastatic and nonmetastatic murine mammary tumor cells. Cancer Res 49: 4724–4728, 1989
Weithmann KU, Peterson H, Sevanian A: Incorporation of arachidonic, dihomogammalinolenic and eicosapentaenoic acids into cultured V79 cells. Lipids 24: 173–178, 1989
Marra CA, deAlaniz MJT, Brenner RR: Uptake and metabolism of eicosa-8, 11,14-trienoic acid in normal hepatocytes and HTC cells. Mol Cell Biochem 65: 143–152, 1984
Chapkin RS, Coble KJ: Utilization of gammalinolenic acid by mouse peritoneal macrophages. Biochim Biophys Acta 1085: 365–370, 1991
Cook HW, Spence MW: Triacylglycerol as a precursor in phospholipid biosynthesis in cultured neuroblastoma cells: studies with labeled glucose, fatty acid, and triacylglycerol. Can J Biochem Cell Biol 63: 919–926, 1985
Dunbar LM, Bailey JM: Enzyme delections and essential fatty acid metabolism in cultured cells. J Biol Chem 250: 1152–1153, 1974
Brenner RR, Peluffo RO: Regulation of unsaturated fatty acids biosynthesis I. Effect of unsaturated fatty acid of 18 carbons on the microsomal desaturation of linoleic acid into γ-linolenic acid. Biochim Biophys Acta 176: 471–479, 1969
Lands WEM, Inoue M, Sugiura Y, Okuyama H: Selective incorporation of polyunsaturated fatty acids into phosphatidylcholine by rat liver microsomes. J Biol Chem 257: 14968–14972, 1982
Stacey WD, Yu C-L, Wei F-S, Tsai M-H: The role of p21 ras in proliferative signal transduction. In: D. Spandidos (ed). ras Oncogenes. Plenum Press, New York, N.Y., 1989, pp 129–139
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Antueno, R.J., Cantrill, R.C., Huang, YS. et al. Metabolism of n-6 fatty acids by NIH-3T3 cells transfected with the ras oncogene. Mol Cell Biochem 139, 71–81 (1994). https://doi.org/10.1007/BF00944205
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00944205