Summary
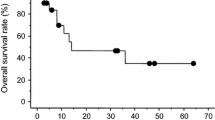
Fifteen patients with advanced, cisplatin-refractory germ cell tumors (GCT) were treated with iproplatin (CHIP). No objective responses were noted in any of the patients treated. By restricting the entry criteria to heavily pre-treated patients, the identification of new active agents in phase II trials may be hindered. Alternative strategies for the investigation of new agents in patients with GCT should be considered, particularly when studying the efficacy and relative toxicity of platinum analogues.
Similar content being viewed by others
References
Lee FH, Canetta R, Issell BF, Lanaz L: New platinum complexes in clinical trials. Cancer Treat Rep 10:29–51, 1983
Creaven PJ, Madajewica S, Pendyala L, Mittleman A, Pontes E, Spaulding M, Arback S, Soloman J: Phase I Clinical Trial of cis-dichloro-trans-dihydroxy-bis-isopropylamine platinum (IV) (CHIP). Cancer Treat Rep 67:795–800, 1983
Miller AB, Hoogstraten B, Staquet M, Winkler A: Reporting results of cancer treatment. Cancer 47:207–214, 1981
Drasga RE, Williams SD, Einhorn LH, Birch R: Phase II Evaluation of iproplatin in refractory germ cell tumors: a Southwestern Cancer Study Group Trial. Cancer Treat Rep 71:863–864, 1987
Claven M, Monfardini S, Siegenthaler P, Pinedo HM, Renard J: Phase II trial of CHIP in previously treated disseminated testicular cancer. Proc ASCO 5:108, 1986
Motzer RJ, Cooper K, Geller NL, Bajorin DF, Dmitrovsky E, Herr H, Morse M, Fair W, Pramod S, Russo P, Bosl GJ: The role of ifosfamide plus cisplatin-based chemotherapy as salvage therapy for patients with refractory germ cell tumors. Cancer 66:2476–2481, 1990
Motzer RJ, Gulati SC, Crown JP, Weisen S, Doherty M, Herr H, Fair W, Sheinfeld J, Sogani P, Russo P, Bosl GJ: High-dose chemotherapy and autologous bone marrow rescue for patients with refractory germ cell tumors. Cancer 69:550–556, 1992
Motzer R, Geller N, Tan C, Herr H, Morse M, Fair W, Sheinfeld J, Sogani P, Russo P, Bosl G: Salvage chemotherapy for patients with germ cell tumors. Cancer 67:1305–1310, 1991
Motzer RJ, Bosl GJ, Tauer K, Golbey R: Phase II trial of carboplatin in patients with advanced germ cell tumors refractory to cisplatin. Cancer Treat Rep 71:197–198, 1987
O'Reilly SM, Rustin GJS, Smith DB, Newlands ES: Single agent activity of carboplatin in patients with previously untreated non-seminomatous germ cell tumors. Annals of Oncology 3:1992 (in press)
Pederson AG, Bork E, Osterling K, Dombernowsky P, Hanson H: Phase II study of teniposide in small cell carcinoma of the lung. Cancer Treat Rep 68:1289–1291, 1984
Bork E, Hansen M, Dombernowsky P, Hansen S, Pederson AG, Hansen HH: Teniposide (VM-26) an overlooked highly active agent in small-cell lung cancer: results of a phase II trial in untreated patients. J Clin Oncol 4:524–527, 1986
Evans WK, Eisenhauer EA, Cormier Y, Wierzbicki R, Laberge F, Shepherd FA: Phase II Study of Amonafide: results of treatment and lessons learned from the study of an investigational agent in previously untreated patients with extensive small-cell lung cancer. J Clin Oncol 8:390–395, 1990
Holmes AF, Walters RS, Theriault RL, Forman AD, Newton LK, Raber MN, Buzdar AU, Frye DK, Hortobagyi GN: Phase II trial of taxol, an active drug in the treatment of metastatic breast cancer. J Natl Cancer Inst 83:1797–1805, 1991
Ettinger DS, Finkelstein DM, Abeloff MD, Bonomi PD: Justification for evaluating new anticancer drugs in selected untreated patients with a chemotherapy-sensitive advanced cancer: an ECOG randomized trial. Proc ASCO 9:864, 1990
Grant SC, Gralla RJ, Kris MG, Orazem J, Kitsis A: Single-agent chemotherapy trials in small-cell lung cancer, 1970–1990: the case for studies in previously treated patients. J Clin Oncol 10:484–498, 1992
Nichols C, Saxman S, Williams S, Loehrer PJ, Miller ME, Wright C, Einhorn LH: Primary mediastinal nonsemenomatous germ cell tumors — a modern single institution experience. Cancer 65:1641–1646, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Murphy, B.A., Motzer, R.J. & Bosl, G.J. Phase II study of iproplatin (CHIP) in patients with cisplatin-refractory germ cell tumors; the need for alternative strategies in the investigation of new agents in GCT. Invest New Drugs 10, 327–330 (1992). https://doi.org/10.1007/BF00944190
Issue Date:
DOI: https://doi.org/10.1007/BF00944190