Abstract
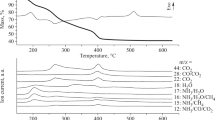
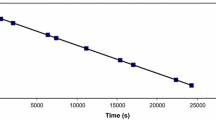
The kinetics of solid-phase reactions of substitution of coordinated ammonia by outer-sphere bromide ions in [CrL(NH3)2]2Br3·nH2O, where L is 1,4,8,11- tetraazaundecane, n ≠ 2–2.5, was studied by a nonisothermal method by differential thermogravimetry, mass spectrometry, and investigations in a flow-type reactor. The kinetic stability of the isomeric salts increases in the series trans<cis-β<cis-d. Tetragonal distortion of the chromophore is of decisive significance for the trans isomer, while the difference in the stability of the cis isomers is related to the structure of their crystal lattices.
Similar content being viewed by others
Literature cited
Yu. N. Shevchenko, E. A. Pisarev, and K. B. Yatsimirskii, “Synthesis and properties of isomeric hexaammine mixed-ligand complex salts of chromium(III) bromide with 1,4,8,11-tetraazaundecane and ammonia,” Zh. Obshch. Khim.,55. No. 12, 2788–2793 (1985).
V. A. Logvinenko, Thermal Analysis of Coordination Compounds and Clathrates [In Russian], Nauka, Novosibirsk (1982).
A. V. Nikolaev, V. A. Logvinenko, Ya. Shestak, and V. Shkvar, “Study of thermal solidphase transformations of coordination compounds by nonisothermal kinetic methods,” Dokl. Akad. Nauk SSSR,231, No. 1, 146–149 (1976).
V. A. Nazarenko, Yu. N. Shevchenio, and É. N. Korol', “Kinetics of solid-phase ligandsubstitution reactions in chromium(III) complexes,” in: Second Symposium “Kinetic Mass Spectrometry“: Abstracts of Papers, Moscow, Oct. 9–11, 1984, Chernogolovka (1984), p. 24.
Author information
Authors and Affiliations
Additional information
Translated from Teoreticheskaya i Éxperimental'naya Khimiya, Vol. 21, No. 5, pp. 637–639, September–October, 1985.
Rights and permissions
About this article
Cite this article
Pisarev, E.A., Shevchenko, Y.N., Logvinenko, V.A. et al. Kinetics of solid-phase ligand-substitution reactions in diammine(1,4,8,11-Tetraazaundecane)chromium(III) bromide isomers. Theor Exp Chem 21, 610–612 (1985). https://doi.org/10.1007/BF00944106
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00944106