Abstract
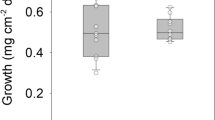
Very little information exists on the effects of nitrate on corals, although this is the major form in which nitrogen is prescrit in tropical eutrophie coastal waters. In this study we incubated nubbins ofPorites porites and explants ofMontastrea annularis in laboratory photostats illuminated by halide lamps, with concentrations of nitrate of 0, 1, 5 and 20 μM, for 40 and 30 d, respectively, At the end of this period it was found that the population density of the zooxanthellae had increased significantly with increased nitrate concentration, suggesting nitrogen limitation of the growth rate of zooxanthellae in the control group. There were also significant increases in the amount of chlorophylla ande 2 per algal cell, in the volume of the algal cells, and in the protein per cell. Overall, the protein per unit surface increased, but this was attributable solely to increased algal protein: there was no significant change in host protein. Maximum gross photosynthesis normalized to surface area was enhanced by nitrate addition, reflecting the increase in algal population density. There was no change when normalized on a per cell basis. Respiration rate normalized to protein content was decreased by nitrate. The most dramatic change was in the rate of skeletogenesis, which decreased by ≅ in both species when exposed to nitrate enrichment. A model is presented which suggests that the diffusion-limited supply of CO2 from surrounding seawater is used preferentially by the enlarged zooxanthellae population for Photosynthesis, thereby reducing the availability of inorganic carbon for calcification. It is concluded that enhanced nitrate levels in tropical coastal waters will have a hitherto unrecognized effect on the growth rate of tropical coral reefs.
Similar content being viewed by others
References
Atkinson MJ, Carlson B, Crow GL (1995) Coral growth in highnutrient, low-pH seawater: a case study of corals raised in the Waikiki Aquarium. Coral Reefs 14: 215–223
Berner T, Izhaki I (1994) Effect of exogcnous nitrogen levels on ultrastructure of zooxanthellae from the hermatypic coxalPocillopora damicornis. Pacif Sci 48: 254–262
Burris JE, Porter JW, Laing WA (1983) Effects of carbon dioxide concentration on coxal photosynthesis. Mar Biol 75: 113–116
Bythell JC (1990) Nutrient uptake in the reef-building coral Acropora palmata at natural environmental concentrations. Mar Ecol Prog Ser 68: 65–69
Coffroth MA (1991) Cyclical mucous sheet formation on poritid corals in the San Blas Islands, Panama. Mar Biol 109: 35–40
Cook CB, D'Elia CF, Muller-Parker G (1988) Host feeding and nutrient sufficiency for zooxanthellae in the sea anemoneAiptasia pallida. Mar Biol 98: 253–262
Crossland CJ, Barnes DJ (1977) Nitrate assimilation enzymes from two hard corals,Acropora acuminata andGoniastrea australensis. Comp Biochem Physiol 57B: 151–157
Davies PS (1984) The role of zooxanthellae in the nutritional energy requirements ofPocillopora eydouxi. Coral Reefs 2: 181–186
Davies PS (1989) Short-term growth measurements of corals using an accurate buoyant weighing technique. Mar Biol 101: 389–395
Davies PS (1995) Coral nubbins and explants for reef assessment and laboratory ecotoxicology. Coral Reefs 14: 267–269
D'Elia CF, Webb KL (1977) The dissolved nitrogen flux of reef corals. Proc 3rd int coxal Reef Symp 1: 325–330 [Taylor DL (ed) Rosenstiel School of Marine and Atmospheric Science, University of Miami]
Dennison WC, Barnes DJ (1988) Effect of water motion on coxal photosynthesis and calcification. J exp mar Biol Ecol 115: 67–77
Doty MS (1971) Measurement of water movement in reference to algal benthic growth. Botanica mar 14: 32–35
Dubinsky Z, Stambler N, Ben-zion M, McCloskey LR, Muscatine L, Falkowsky PG (1990) The effect of external nutrient resources on the optical properties and photosynthetic efficiency of Stylophora pistillata. Proc R Soc (Ser B) 239: 231–246
Franzisket L (1974) Nitrate uptake by reef corals. Int Revue ges Hydrobiol 59: 1–7
Harland AD, Davies PD (1995) Symbiont photosynthesis increases both respiration and photosynthesis in the symbiotic sea anemoneAnemonia viridis. Mar Biol 123: 715–722
Hawker DW, Connell DW (1992) Standards and criteria for pollution control in coxal reef areas. In: Connell DW, Hawker DW (eds) Pollution in tropical aquatic systems. CRC Press, New York, pp 169–191
Hoegh-Guldberg O, Smith GJ (1989) Influence of the population density of zooxanthellae and supply of ammonium on the biomass and metabolic characteristics of of the reef coralsSeriatophora hystrix andStylophora pistillata. Mar Ecol Prog Ser 57: 173–186
Jeffrey SW, Humphxey GF (1975) New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem Physiol Pfl 167: 191–194
Kinsey DW (1987) Responses of coxal reef systems to elevated nutrient levels. In: Baldwin CL (ed) Nutrients in the Great Barrier Reef Region. Great Barrier Reef Marine Park Authority, Australia, pp 55–65
Kinsey DW, Davies PI (1979) Effects of elevated nitrogen and phosphorus on coxal reef growth. Limnol Oceanogr 24: 935–940
Lesser MP, Weis VM, Patterson MR, Jokiel PL (1994) Effects of morphology and water motion on carbon delivery and productivity in the reef coral,Pocillopora damicornis (Linnaeus): diffusion barriers, inorganic carbon limitation, and biochemical plasticity. J exp mar Biol Ecol 178: 153–179
Lowry OH, Roscbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J biol Chem 193: 265–275
Markwell MAK, Haas SM, Bieber LL, Tolbert NE (1978) A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Analyt Biochem 87: 206–210
Miller DJ, Yellowlees D (1989) Inorganic nitrogen uptake by symbiotic marine cnidarians: a critical review. Proc R Soc (Ser B) 237: 109–125
Muller-Parker G, Cook CB, D'Elia CF (1994a) Elemental composition of the coralPocillopora damicornis exposed to elevated seawater ammonium. Pacif Sci 48: 234–246
Muller-Parker G, McCloskey LR, Hoegh-Guldberg O, McAuley PJ (1994b) Effect of ammonium enrichment on animal and algal biomass of the coxalPocillopora damicornis. Pacif Sci 48: 273–283
Muscatine L (1967) Glycerol excretion by symbiotic algae from corals and tridacna and its control by the host. Science, NY 156: 516–519
Muscatine L, Falkowski PG, Dubinsky Z, Cook PA, McCloskey LR (1989) The effect of external nutrient resources on the population dynamics of zooxanthellae in a reef coxal. Proc R Soc (Ser B) 236: 311–324
Muscatine L, Falkowsky PG, Porter JW, Dubinsky Z (1984) Fate of photo synthetically fixed carbon in light- and shade-adapted colonies of the symbiotic coralStylophora pistillata. Proc R Soc (Ser B) 222: 181–202
Prézelin BB (1987) Photosynthetic physiology of dinoflagellates. In: Taylor FJR (ed) The biology of dinoflagellates. Blackwell Scientific Publishers, Oxford, pp 174–223
Rinkevich B, Loya Y (1983) Oriented translocation of energy in grafted reef corals. Coral Reefs 1: 243–247
Snidvongs A, Kinzie III RA (1994) Effects of nitrogen and phosphorus enrichment on in vivo symbiotic zooxanthellae ofPocillopora damicornis. Mar Biol 118: 705–711
Stambler N, Popper N, Dubinsky Z, Stimson J (1991) Effects of nutrient enrichment and water motion on the coralPocillopora damicornis. Pacif Sci 45: 299–307
Stimson J (1992) The effect of ammonium addition on coxal growth rate. Proc. 7th int coral Reef Symp (Abstract) 1: p. 383 [Richmond RH (ed) University of Guam, Mangilao, Guam]
Stimson J, Kinzie RA (1991) The temporal pattern and rate of release of zooxanthellae from the reef coralPocillopora damicornis (L.) under nitrogen-enrichment and control conditions. J exp mar Biol Ecol 153: 63–74
Strickland JDH, Parsons TR (1972) A practical handbook of seawater analysis. 2nd edn. Bull Fish Res Bd Can 167: 1–310
Syrett PJ (1981) Nitrogen metabolism of microalgae. Physiological bases of phytoplankton ecology. Can Bull Fish aquat Sciences 210: 182–210
Szmant AM, Ferrer LM, FitzGerald LM (1990) Nitrogen excretion and O:N ratios in reef corals: evidence for conservation of nitrogen. Mar Biol 104: 119–127
Tomascik T, Sander F (1985) Effects of eutrophication on reefbuilding corals. I. Growth rate of the reef-building coralMontastrea annularis. Mar Biol 87: 143–155
Walter SD, Feinstein AR, Wells CK (1987) Coding ordinal independent variables in multiple regression analyses. Am J Epidem 125: 319–323
Webb KL, Wiebe WJ (1978) The kinetics and possible significance of nitrate uptake by several algal-invertebrate symbioses. Mar Biol 47: 21–27
Weis VM, Smith GJ, Muscatine L (1989) A “CO2 supply” mechanism in zooxanthellate cnidarians: role of carbonic anhydrase. Mar Biol 100: 195–202
Wilkerson FP, Kremer P (1992) DIN, DON, PO4 flux by a medusa with algal symbionts. Mar Ecol Prog Sex 90: 237–250
Wilkerson FP, Muscatine L (1984) Uptake and assimilation of dissolved inorganic nitrogen by a symbiotic sea anemone. Proc R Soc (Sex B) 221: 71–86
Wilkerson FP, Trench RK (1986) Uptake of dissolved inorganic nitrogen by the symbiotic clamTridacna gigas and the coralAcropora sp. Mar Biol 93: 237–246
Zar JH (1984) Biostatistical analysis. 2nd edn. Prentice-Hall International, Inc., Englewood Cliffs, New Jersey
Author information
Authors and Affiliations
Additional information
Communicated by J.P. Thorpe, Port Erin F. Marubini
Rights and permissions
About this article
Cite this article
Marubini, F., Davies, P.S. Nitrate increases zooxanthellae population density and reduces skeletogenesis in corals. Mar. Biol. 127, 319–328 (1996). https://doi.org/10.1007/BF00942117
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00942117