Abstract
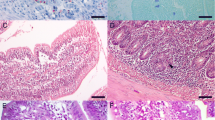
Groups of five rats each were infected with metacercariae ofFasciola hepatica according to two experimental procedures. In the first experiment, they received 20 metacercariae 0, 1, 2 or 3 times at intervals of 4 weeks. In the second, both the inoculation dose and the interval between doses were varied. After the animals had been killed, the small intestine was prepared for histology and tissue sections were stained for the detection and quantification of mucosal mast cells (MMCs), eosinophils and goblet cells. The density of eosinophils and MMCs increased significantly with each reinfection in the first experiment, in the second, the MMC reaction reached a peak at 4 weeks post-infection (p.i.), whereas the eosinophils peaked at 4 and 17 weeks p.i. In addition, the reaction of these cells depended on the infectious dose. In both experiments, goblet cells reacted only weakly. These findings thus clearly reflect a considerable intestinal response against migrating juvenileF. hepatica. The pattern of these defence reactions in the small intestine suggests some similarities with the mechanisms of antibody-dependent cell-mediated cytotoxicity that have been shown to take place duringSchistosoma mansoni reinfection.
Similar content being viewed by others
References
Alizadeh H, Wakelin D (1982) Comparison of rapid expulson ofTrichinella spiralis in mice and rats. Int J Parasitol 12:65–73
Bexter A (1982) “Roulade”-Technik/“Swiss roll”-Technik. Histo 1:12–13
Burden DJ, Hughes DL, Hammet NC (1982)Fasciola hepatica: antibody coating of juvenile flukes in the intestinal lumen of resistant rats. Res Vet Sci 32:44–47
Capron A, Dessaint JP, Haque A, Capron M (1982) Antibodydependent cell-mediated cytotoxicity against parasites. Prog Allergy 31:234–267
Davies C, Goose J (1981) Killing of newly excysted juveniles ofFasciola hepatica in sensitized rats. Parasite Immunol 3:81–96
Doy TG, Hughes DL, Harness E (1978) Resistance of the rat to reinfection withFasciola hepatica and the possible involvement of intestinal eosinophil leucocytes. Res Vet Sci 25:41–44
Doy TG, Hughes DL, Harness E (1981) Hypersensitivity in rats infected withFasciola hepatica: possible role in protection against a challenge infection. Res Vet Sci 30:360–363
Doyle JJ (1971) Acquired immunity to experimental infection withFasciola hepatica in cattle. Res Vet Sci 12:527–534
Doyle JJ (1973) Homocytotropic antibodies induced in calves by infection withFasciola hepatica. Int Arch Allergy Appl Immunol 45:744–751
Duffus WPH, Franks D (1980) In vitro effect of immune serum and bovine granulocytes on juvenileFasciola hepatica. Clin Exp Immunol 41:430–440
Enerbäck L (1981) The gut mucosal mast cell. Monogr Allergy 17:222–232
Haig DM, McKee TA, Jarrett EEE, Woodbury RG, Miller HRP (1982) Generation of mucosal mast cells is stimulated in vitro by factors derived from T cells of helminth-infected rats. Nature 300:188–190
Harness E, Hughes DL, Doy TG (1976) The demonstration of pre-hepatic immune responses toFasciola hepatica in the mouse. Int J parasitol 6:15–17
Haroun EM, Hammond JA, Sewell MMH (1980) Resistance toFasciola hepatica in rats and rabbits following sensitising infection and treatment. Res Vet Sci 28:377–379
Hayes TJ, Bailer J, Mitrovic M (1972) Immunity in rats to superinfection withFasciola hepatica. J Parasitol 58:1103–1105
Jarrett EEE, Haig DM (1984) Mucosal mast cells in vivo and in vitro. Immunol Today 5:115–118
Jarrett EEE, Miller HRP (1982) Production and activities of IgE in helminth infections. Prog Allergy 31:178–233
Lendrum AC (1944) The staining of eosinophil polymorphs and enterochromaffin cells in histological sections (abstract). J Pathol Bacteriol 56:441
Luna LG (1968) Methods for cytoplasmic granules. In: Luna LG (ed) Manual of histologic staining methods of the Armed Forces Institute of Pathology (AFIP). AFIP, Washington, DC, pp 111–112
Mahmoud AAF (1980) Eosinophilopoiesis. In: Mahmoud AAF, Austen KF (eds) The eosinophil in health and disease. Grune & Stratton, New York, pp 61–75
Meierhofer B (1987) Zelluläre Reaktionen im Dünndarm von Ratten nach Erst- bzw. Reinfektion mitFasciola hepatica. Thesis, Faculty of Veterinary Medicine, University of Berm, Bern
Milbourne EA, Howell MJ (1990) Eosinophil responses toFasciola hepatica in rodents (Abstract). Bull Soc Fr Parasitol 8 [Suppl 1]:632
Miller HRP (1984) The protective mucosal response against gastrointestinal nematodes in ruminants and laboratory animals. Vet Immunol Immunopathol 6:167–259
Miller HRP, Nawa Y (1979)Nippostrongylus brasiliensis: intestinal goblet cell response in adoptively immunized rats. Exp Parasitol 47:81–90
Moraes FR, Silva VMS, Vasconcelos OT, Costa AJ, Moraes JRE (1986) Mastócitos da mucosa intestinal x carga parasitária em suínos naturalmente infectados por nematóides. Ars Vet 2:255–262
Oldham G (1985) Immune responses in rats and cattle to primary infections withFasciola hepatica. Res Vet Sci 39:357–363
Palmer DG (1984) Die Entwicklung der nicht-epithelialen Zellpopulationen in der Darmschleimhaut des neonatalen Kalbes im Hinblick auf die Ausbildung des Schleimhautimmunsystems. Thesis, Raculty of Veterinary Medicine, University of Zurich, Zurich
Pfister K (1984) Role of IgE in fascioliasis. Prev Vet Med 2:359–370
Pfister K (1986) Seroepidemiological and immunological studies on fascioliasis. Habilitation thesis, Faculty of Veterinary Medicine, University of Bern, Bern
Pfister K, Meierhofer B (1986) Cellular responses in the small intestine and liver ofFasciola hepatica-infected rats. Mitt Oesterr Ges Tropenmed Parasitol 8:73–82
Pfister K, Turner K, Currie A, Hall E, Jarrett EEE (1983) IgE production in rat fascioliasis. Parasite Immunol 5:587–593
Smith NC, Bryant C, Ovington VS (1990) Responses of nude rats to infection withNippostrongylus brasilientsis (Abstract). Bull Soc Fr Parasitol 8 [Suppl 1]:591
Wedrychowicz H, Turner K, Pfister K, Holmes PH, Armour J (1984) Local antibody responses in the bile and faeces of sheep infected withFesciola hepatica. Res Vet Sci 37:44–51
Weibel ER (1979) Stereological methods 1. Practical methods for biological morphometry, vol 1. Academic Press, London
Woodbury RG, Miller HRP, Huntley JF, Newlands GFJ, Palliser AC, Wakelin D (1984) Mucosal mast cells are functionally active during spontaneous expulsion of intestinal nematode infections in rat. Nature 312:450–452
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. J. Eckert (Zürich) on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Charbon, J.L., Spähni, M., Wicki, P. et al. Cellular reactions in the small intestine of rats after infection withFasciola hepatica . Parasitol Res 77, 425–429 (1991). https://doi.org/10.1007/BF00931639
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00931639