Abstract
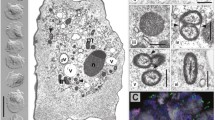
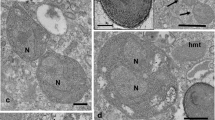
The isolation of two plasmind-like ciruclar DNAs, measuring 52 and 42 kbp, from anAcanthamoeba sp. from the Philippines has led to the demonstration of a bacterial endosymbiont occurring in this free-living amoeba. The 52-kbp band hybridized with a short sequence of cytochrome b gene and was identified as the mitochondrial DNA, whereas the 42-kbp band was identified as plasmid DNA of the bacterial symbionts on the basis of electron microscopy. The endosymbionts are gram-negative, rod-shaped bacteria measuring approximately 1.3×0.43 μm and numbering about eight to ten cells per section. They are randomly distributed in both cysts and trophozoites and are surrounded neither by a phagolysosomal membrane nor by a clear or electrontranslucent region. The endosymbiont membrane appears to have a close association with ribosomes, which are seen to be more concentrated within the vicinity of the symbionts than elsewhere within the cytoplasm. Attempts to grow the symbionts and the amoebae separately have failed.
Similar content being viewed by others
References
Barbaree JM, Fields BS, Feeley JC, Gorman GW, Martin WT (1986) Isolation of protozoa from water associated with a legionellosis outbreak and demonstration of intracellular multiplication ofLegionella pneumophila. Appl Environ Microbiol 51:422–424
Bogler SA, Zarley CD, Burianek LL, Fuerst PA, Byers TJ (1983) Interstrain mitochondrial DNA polymorphism detected inAcanthamoeba by restriction endonuclease analysis. Mol Biochem Parasitol 8:145–163
Chavez LA, Balamuth W, Gong T (1986) A light and electron microscopical study of a new, polymorphic free-living amoeba,Phreatamoeba balamuthi n.g., n. sp. J Protozool 33:397–404
Costas M, Edwards SW, Lloyd D, Griffiths AJ, Turner G (1983) Restriction enzyme analysis of mitochondrial DNA of members of the genusAcanthamoeba as an aid in taxonomy. FEMS Microbiol Lett 17:231–234
Drozanski WJ (1991)Sarcobium lytium gen. nov., sp. nov., an obligate intracellular bacterial parasite of small free-living amoebea. Int J Syst Bacteriol 41:82–87
Fields BS, Sanden GN, Barbaree JM, Morrill WE, Wadowsky RM, White EH, Feely JC (1989) Intracellular multiplication ofLegionella pneumophila in amoebae isolated from hospital hot water tanks. Curr Microbiol 18:131–137
Fritsche TR, Gauton RK, Seyedirashti S, Bergeron DL, Lindquist TD (1983) Oceurrence of bacterial endosymbionts inAcanthamoeba spp. isolated from corneal and environmental specimens and contact lenses. J Clin Microbiol 31:1122–1126
Gautom RK, Lory S, Seyedidrashti S, Bergeron DL, Fritsche TR (1994) Mitochondrial DNA fingerprinting ofAcanthamoeba spp. isolated from clinical and environmental sources. J Clin Microbiol 32:1070–1073
Hall J, Voelz H (1985) Bacterial endosymbionts ofAcanthamoeba sp. J Parasitol 71:89–95
Gortz HD (1982) Infection ofParamecium bursaria with bacteria and yeast. J Cell Sci 58:445–453
Jeon KW, Lorch IJ (1967) Unusual intracellular bacterial infection in large, free-living amoebae. Exp Cell Res 48:236–240
Marten PA, Clayton DA (1979) Mechanism of mitochondrial DNA replication in mouse L-cell: localization and sequence of the light-strand origin of replication. J Mol Biol 135:327–351
McLaughin GL, Brandt FH, Visvesvara GS (1988) Restriction fragment length polymorphisms of the DNA of selectedNaegleria andAcanthamoeba amoebae. J Clin Microbiol 26:1655–1658
Page FC (1988) A new key to freshwater and soil gymnamoebae. Serry House, Ambleside, UK
Phillips BP (1974)Naegleria: another pathogenic ameba. Studies in germfree guinea pigs. Am J Trop Med Hyg 23:850–855
Preer JR Jr, Preer LB, Jurand A (1974) Kappa and other endosymbionts inParamecium aurelia. Bacteriol Rev 38:113–163
Proca-Ciobanu M, Lupascu GH, Pertrovici AL, Ionescu MD (1975) Electron microscopic study of a pathogenicAcanthamoeba castellani strain: the presence of bacterial symbionts. Int J Parasitol 5:49–56
Radulescu S, Meyer EA, Burghela B, Meitert T (1988) Ultrastructural study of a bacterial symbiont ofGiardia lamblia. In: Wallis PM, Hammond BP (eds) Advances inGiardia research. University of Calgary Press, Alberta, Canada
Rowbotham LJ (1980) Preliminary report on the pathogenicity ofLegionella pneumophila for freshwater and soil amoebae. J Clin Pathol 33:1179–1183
Springer N, Ludwig W, Drozanski W, Amann R, Schleifer KH (1992) The phylogenetic status ofSarcobium lyticum, an obligate intracellular bacterial parasite of small amoebae. FEMS Microbiol Lett 96:199–202
Yagita K (1993) Characterization ofAcanthamoeba isolates from eye infections and the environment by restriction endonuclease digestion of mitochondrial DNA. (in Japanese, with English abstract). Jpn J Parasitol 42:468–478
Yagita K, Endo T (1990) Restriction enzyme analysis of mitochondrial DNA ofAcanthamoeba strains in Japan. J Protozool 37:570–575
Yamamoto H, Sugiura M, Kusunoki S, Ezaki T, Ikeda M, Yabuuchi E (1992) Factors stimulating propagation ofLegionella in cooling tower water. Appl Environ Microbiol 58:1394–1397
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yagita, K., Matias, R.R., Yasuda, T. et al. Acanthamoeba sp. from the Philippines: electron microscopy studies on naturally occurring bacterial symbionts. Parasitol Res 81, 98–102 (1995). https://doi.org/10.1007/BF00931612
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00931612