Abstract
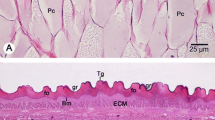
The effect on the tegument of adultFasciola hepatica of incubation in the sodium ionophore monensin, the Na+/K+-ATPase inhibitor ouabain and ouabain pretreatment followed by monensin has been determined in vitro by scanning and transmission electron microscopy (SEM, TEM). With monensin incubation alone (1×10−6 M), a flattening of the tegument with some loss of spines on the ventral surface is evident from 0.5 h onwards. Internally, the subtegumental musculature becomes grossly swollen, although there is no swelling of the infoldings of the basal plasma membrane of the tegument, even after 24 h incubation. Ouabain incubation (1×10−3 M) induces folding of the apical surface of the tegument from 0.5 h onwards, and this is accompanied by the formation of bleds and microvilli. Brief (0.5 h) exposure to ouabain (1×10−3 M) followed by monensin treatment (1×10−4 M, 3 h) leads to gross “vacuolation” of the tegument, but this is not due to swelling of the basal infoldings. The other main feature of ouabain-pretreated flukes is the projection of basal lamina-like material into the tegumental syncytium. Monensin treatment alone (1×10−6 M) results in the Golgi complexes of the tegumental cells becoming very diffuse from 1.5 h onwards, and relatively few secretory bodies are present in the cytoplasm. After 0.5 h incubation in ouabain (1×10−3 M), the Golgi complexes of the tegumental cells are indistinct, although numerous secretory bodies are still present. The classical monensin-induced swelling of the Golgi cisternae is observed in the tegumental cells only when monensin treatment (1×10−4 M, 3 h) was preceded by brief (0.5 h) exposure to ouabain (1×10−3 M). The results are discussed in relation to the postulated osmoregulatory role of the tegument and the role of sodium pumps in membrane function in the fluke.
Similar content being viewed by others
References
Bennett CE, Hughes DL, Harness E (1980)Fasciola hepatica: changes in tegument during killing of adult flukes surgically transferred to sensitised rats. Parasite Immunol 2:39–55
Brock TA, Smith JB (1982) Reversible stimulation of the Na+/K+ pump by monensin in cultures of vascular smooth muscle. Life Sci 31:1043–1050
Brock TA, Lewis LJ, Smith JB (1982) Angiotensin increases Na+ entry and Na+/K+ pump activity in cultures of smooth muscle from rat aorta. Proc Natl Acad Sci USA 79:1438–1442
Dunphy WG, Rothman JE (1985) Compartmental organization of the Golgi stack. Cell 42:13–21
Ellinger A, Pavelka M (1984) Effect of monensin on the Golgi apparatus of absorptive cells in the small intestine of the rat. Morphological and cytochemical studies. Cell Tissue Res 235:187–194
Fahim M, Mangel A, Pressman BC (1983) Effects of ionophore-mediated transport on the cardiac resting potential. J Exp Biol 107:491–493
Fairweather I, Skuce PJ (1985) An evaluation of the fasciolicidal potential of monensin. I. The effect of monensin on the neuromuscular and osmoregulatory systems ofFasciola hepatica. Proceedings of the British Society for Parasitology, Spring Meeting held at the University of Nottingham, March 1985, p 59. Parasitology 91:420
Fairweather I, Anderson HR, Threadgold LT (1986)Fasciola hepatica: tegumental changes in the adult fluke induced by the deacetylated (amine) metabolite of diamphenethide in vitro. Exp Parasitol 62:336–348
Fairweather I, Anderson HR, Threadgold LT (1988a)Fasciola hepatica: morphological changes in vitelline cells following treatment in vitro with the deacetylated (amine) metabolite of diamphenethide (DAMD). Int J Parasitol (in press)
Fairweather I, Skuce PJ, Holmes SD (1988b) Diamphenethide — a reassessment of its pharmacological action. Parasitol Res 74:456–462
Haney ME, Hoehn MM (1968) Monensin, a new biologically active compound. I. Discovery and isolation. Antimicrob Agents Chemother 1967:349–352
Hanna REB (1980)Fasciola hepatica: an immunofluorescent study of antigenic changes in the tegument during development in the rat and the sheep. Exp Parasitol 50:155–170
Ledger PW, Tanzer ML (1984) Monensin — a perturbant of cellular physiology. TIBS 9:313–314
Ozaki H, Kishimoto T, Karaki H, Urakawa N (1982) Effects of the Na inonophore monensin on the contractile response and the movements of monovalent cations in the vascular smooth muscle of rabbit aorta. Naunyn-Schmiedebergs Arch Pharmacol 321:140–144
Raun AP, Cooley CO, Potter EL, Rathmacher RP, Richardson LF (1976) Effect of monensin on feed efficiency of feedlot cattle. J Anim Sci 43:670–677
Rew RS, Fetterer RH, Martin TC (1983)Fasciola hepatica: effects of diamfenetide free amine on in vitro physiology, biochemistry and morphology. Exp Parasitol 55:159–167
Rogan MT, Richards KS (1986)Echinococcus granulosus: in vitro effect of monensin on the tegument of the protoscolex. Parasitology 93:347–355
Rogan MT, Threadgold LT (1984)Fasciola hepatica: lectin binding and tegumental alterations. Exp Parasitol 57:248–260
Shumard RF, Callender ME (1968) Monensin a new biologically active compound. VI. Anticoccidial activity. Antimicrob Agents Chemother 1967:369–377
Silk MH, Spence IM (1969) Ultrastructural studies of the blood fluke —Schistosoma mansoni. II. The musculature. S Afr J Med Sci 34:11–20
Skuce PJ, Fairweather I (1985) The effect of monensin on vitelline cell ultrastructure and egg production inFasciola hepatica and the results of efficacy studies in the laboratory rat. Proceedings of the British Society for Parasitology, Spring Meeting held at the University of Nottingham, March 1985, p 59. Parasitology 91:420
Skuce PJ, Fairweather I (1988)Fasciola jepatica: perturbation of secretory activity in the vitelline cells by the sodium ionophore monensin. Exp Parasitol 65:20–30
Skuce PJ, Anderson HR, Fairweather I (1987) The interaction between the deacetylated (amine) metabolite of diamphenethide (DAMD) and cytochemically demonstrable Na+/K+-ATPase activity in the tegument ofFasciola hepatica. Parasitol Res 74:161–167
Smith JB, Rozengurt E (1978) Serum stimulates the Na+, K+ pump in quiescent fibroblasts by increasing Na+ entry. Proc Natl Acad Sci USA 75:5560–5564
Spurr AR (1969) A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–43
Tartakoff AM (1982) The Golgi complex: crossroads for vesicular traffic. Int Rev Exp Pathol 22:227–251
Tartakoff AM (1982) Simplifying the Golgi complex. TIBS 7:174–176
Tartakoff AM (1983a) Perturbation of vesicular traffic with the carboxylic ionophore monensin. Cell 32:1026–1028
Tartakoff AM (1983b) Perturbation of the structure and function of the Golgi complex by monovalent carboxylic ionophores. Methods Enzymol 98:47–59
Threadgold LT (1985)Fasciola hepatica: interaction of the tegument with poly-L-lysine and enzymes. Exp Parasitol 59:222–230
Threadgold LT, Brennan GP (1978)Fasciola hepatica: basal infolds and associated vacuoles of the tegument. Exp Parasitol 46:300–316
Van Vleet JF, Ferrans VJ (1984a) Ultrastructural alterations in the atrial myocardium of pigs with acute monensin toxicosis. Am J Pathol 114:367–379
Van Vleet JF, Ferrans VJ (1984b) Ultrastructural alterations in skeletal muscle of pigs with acute monensin myotoxicosis. Am J Pathol 114:461–471
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Skuce, P.J., Fairweather, I. Fasciola hepatica: the effect of the sodium inophore monensin on the adult tegument. Parasitol Res 75, 223–232 (1989). https://doi.org/10.1007/BF00931280
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00931280