Abstract
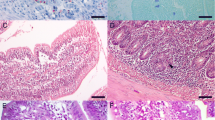
Primary egg-derived infection ofHymenolepis nana (100 eggs) in BALB/c (rapid responder) and C3H (slow responder) mice resulted in increased levels of mucosal mast cells (MMCs), eosinophilia (bone marrow, peripheral, tissue) and phospholipase B activity. The response appeared to be similar in both strains used, with a slight difference in cellular accumulation but a significantly earlier response in BALB/c than in C3H mice. These findings suggest that the prolongation ofH. nana infection in C3H mice may be related to the delayed appearance of MMCs and eosinophils, which triggers a slower generation of the intestinal inflammation response. The rapidity with which phospholipase B activity increased was strictly correlated with eosinophil tissue number; this further supports the hypothesis for a direct parallel between eosinophils and phospholipase B activity in infected tissue.
Similar content being viewed by others
References
Alizadeh H, Murrell KD (1984) The intestinal mast cell response toTrichinella spiralis infection in mast cell-deficient W/Wv mice. J Parasitol 70:767–773
Alizadeh H, Wakelin D (1982) Comparison of rapid expulsion ofTrichinella spiralis in mice and rats. Int J Parasitol 12:65–73
Asano K, Nakamura F, Okamoto K (1982) Passive protection of nude mice with lymphocytes from mice infected withHymenolepis nana eggs. Jpn J Parasitol 31:391–400
Askenase PW (1980) Jnimunopathology of parasitic disease: involvement of basophils and mast cells. Springer Semin Immunopathol 2:1–59
Bailey WS (1951) Host-tissue reaction to initial and superimposed infections withHymenolepis nana var.fraterna. J Parasitol 37:440–444
Bortoletti G, Conchedda M, Ferretti G (1985) Damage and early destruction ofTaenia taeniaeformis larvae in resistant hosts, and anomalous development in susceptible hosts: a light microscopic and ultrastructural study. Int J Parasitol 15:377–384
Butterworth AE (1977) The eosinophil and its role in immunity to helminth infection. Curr Top Microbiol Immunol 77:127–168
Capron M, Capron A, Torpier G, Bazin H, Bout D, Joseph M (1978) Eosmophil-dependent cytotoxicity in rat schistosomiasis. Involvement of IgG 2a antibody and the role of mast cells. Eur J Immunol 8:127–133
Cheema KJ, Scofield AM (1985) The influence and level of infection of rats withNippostrongylus brasiliensis on the haematology and phospholipase activity and mast cell numbers in the small intestine and colon. Int J Parasitol 15:55–60
Coleman EJ, De Salva JJ (1963) Mast cell responses to cestode infection. Proc Soc Exp Biol Med 112:432–434
Cook RW, Williams JF (1981) Pathology ofTaeniae taeniaeformis infection in the rat: gastrointestinal changes. J Comp Pathol 91:205–216
Dobson C (1972) Immune response to gastrointestinal helminths. In: Soulsby EJL (ed) Immunity to animal parasite. Academic Press, New York, pp 191–222
Duncan DB (1955) Multiple range and multiple F test. Biometrics 11:1–45
Elson CO, Kagnoff MF, Fiocchi C, Befus AD, Targan S (1986) Intestinal immunity and inflammation: recent progress. Gastroenterology 91:746–768
Enerback L (1966) Mast cells in rat gastrointestinal mucosa: 2. Dye binding and metachromatic properties. Acta Pathol Microbiol Scand 66:303–312
Ferretti G, Gabriele F, Palmas C (1980) Methodology in experimental infections of mice withHymenolepis nana. Boll Zool 47:165–184
Ferretti G, Gabriele F, Palmas C (1981) Development of human and mouse strain ofHymenolepis nana in mice. Int J Parasitol 11:425–430
Friedberg W, Neas BR, Faulkner DN, Friedberg MH (1967) Immunity toHymenolepis nana: transfer by spleen cells. J Parasitol 53:895–896
Ftiedberg W, Neas BR, Faulkner DN, Congdon CC (1979)Hymenolepis nana intestinal tissue phase in actively immunized mice. J Parasitol 65:61–64
Furukawa T (1983) Protective immunity toHymenolepis nana infection in mice. Acta Med Kinki Univ 8:165–180
Furukawa T, Miyazato T, Okamoto K, Nakay Y (1977) The fine structure of the hatched onchospheres ofHymenolepis nana. Jpn J Parasitol 26:49–62
Furukawa T, Niwa A, Miyazato T (1981) Ultrastructural aspects of immune damage toHymenolepis nana onchospheres in mice. Int J Parasitol 11:287–300
Gabriele F, Ecea AR, Wakelin D, Palmas C (1986) Blast cell activity in mice infected withHymenolepis nana, H. diminuta andTrichinella spiralis: in vivo uptake of125IUdR in lymphoid tissues and gut. J Helminthol 60:313–321
Heyneman D (1962) Studies on helminth immunity: IV. Rapid onset of resistance by the white mouse against a challenging infection with eggs ofHymenolepis nana (Cestoda: Hymenolepididae). J Immunol 88:217–220
Inoue T (1984) Histopathological studies of mice actively and passively immunized withHymenolepis nana. Acta Med Kinki Univ 9:191–209
Ito A, Yamamoto M (1976) The mode of active protection againstHymenolepis nana reinfection in mice inoculated with different doses of shell-free eggs. Jpn J Parasitol 25:247–253
Larsh JE Jr, Ottolenghi A, Weatherly NF (1974)Trichinella spiralis: phospholipase in challenged mice and rats. Exp Parasitol 36:299–306
Lee TDG, Wakelin D (1982) The use of host strain variation to assess the significance of mucosal mast cells in the spontaneous cure response of mice to the nematodeTrichuris muris. Jnt Arch Allergy Appl Immunol 67:302–305
Lee TDG, Swieter M, Befus AD (1986) Mast cell responses to helminth infection. Parasitol Today 2:186–191
Miller HRP, Jarrett WFH (1971) Immune reaction in mucous membranes: I. Intestinal mast cell response during helminth expulsion in the rat. Immunology 20:277–288
Miyazato T, Furukawa T, Inoue T (1979) Intestinal pathology associated with primary and secondary infections ofHymenolepis nana in mice. Jpn J Parasitol 28:185–195
Ottolenghi A (1970) The relationship between eosinophilic leukocytes and phospholipase B activity in same rat tissues. Lipids 5:531–538
Ottolenghi A, Pickett JP, Greene WB (1967) Histochemical demonstration of phospholipase B (lysolecithinase) activity in rat tissue. J Histochem Cytochem 14:907–914
Palmas C, Ecca AR, Gabriele F (1984a) Effect of vinblastine on the capacity of mesenteric lymph node cells to transfer immunity againstHymenolepis nana. Microbiol Lett 27:145–149
Palmas C, Wakelin D, Gabriele F (1984b) Transfer of immunity againstHymenolepis nana in mice with lymphoid cells or serum from infected donors. Parasitology 89:287–293
Palmas C, Bortoletti G, Conchedda M, Gabriele F (1986) Immunological memory and lymphoblast-migration in mice infected withHymenolepis nana. Z Parasitenkd 72:397–403
Siebert AE Jr, Good AHR, Simmons JE (1979) Ultrastructural aspects of the host cellular immune response toTaenia crassiceps metacestodes. Int J Parasitol 9:323–331
Wakelin D (1984) Immunity to parasite. How animals control parasite infections. Edward Arnold, London
Woodbury RG, Miller HRP, Huntley JF, Newlands GFJ, Palliser AC, Wakelin D (1984) Micosal mast cells are functionally active during spontaneous expulsion of intestinal nematode infections in rat. Nature 312:450–452
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bortoletti, G., Gabriele, F. & Palmas, C. Kinetics of mast cells, eosinophils and phospholipase B activity in the spontaneous-cure response of two strains of mice (rapid and slow responder) to the cestodeHymenolepis nana . Parasitol Res 75, 465–469 (1989). https://doi.org/10.1007/BF00930974
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00930974