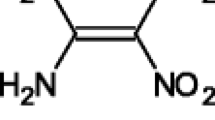
Conclusions
-
1.
A new method was proposed for the preparation of fluorodinitromethane by the electrochemical denitration of fluorotrinitromethane.
-
2.
The electrochemical denitration of fluorotrinitromethane can be run at various cathodes, both those with a high (mercury, graphite) and with a low (platinum) hydrogen overvoltage.
Similar content being viewed by others
Literature cited
S. G. Mairanovskii and V. A. Petrosyan, Elektrokhimiya,2, 115 (1966); V. A. Petrosyan, S. G. Mairanovskii, V. I. Slovetskii, and A. A. Fainzil'berg, Izv. Akad. Nauk SSSR, Ser. Khim., 928 (1968); 2398 (1969); V. A. Petrosyan, V. I. Slovetskii, S. G. Mairanovskii, and A. A. Fainzil'-berg, Elektrokhimiya,6, 1595 (1970).
V. A. Petrosyan, S. G. Mairanovskii, L. V. Okhlobystina, V. I. Slovetskii, and A. A. Fainzil'-berg, Izv. Akad. Nauk SSSR, Ser. Khim., 1690 (1971).
L. V. Okhlobystina, V. M. Khutoretskii, and A. A. Fainzil'berg, ibid., 1487 (1971).
V. I. Slovetskii, L. V. Okhlobystina, A. A. Fainzil'berg, A. I. Ivanov, L. I. Biryukova, and S. S. Novikov, ibid., 2063 (1965).
L. T. Eremenko and F. Ya. Natsibullin, ibid., 912 (1968).
M. Sh. L'vova, V. I. Slovetskii, and A. A. Fainzil'berg, ibid., 649 (1966).
M. Kamlet and H. Adolph, J. Org. Chem.,33, 3073 (1968).
E. Dzhafarov, A. Tomilov, and M. Fioshin, Electrosynthesis of Organic and Inorganic Compounds [in Russian], Azerbaidzhan Gos. Izd., Baku (1965).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 7, pp. 1544–1549, July, 1973.
Rights and permissions
About this article
Cite this article
Petrosyan, V.A., Khutoretskii, V.M., Okhlobystina, L.V. et al. Electrolysis of aliphatic nitro compounds Communication 1. Electrochemical denitration of fluorctrinitromethane. Russ Chem Bull 22, 1497–1501 (1973). https://doi.org/10.1007/BF00930047
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00930047