Conclusions
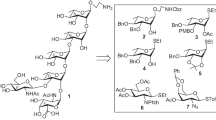
Trisaccharideβ-D-glucopyranosyl-(1→4)-α-L-rhamnopyranosyl-(1→ 3)-D-galactose was synthesized by using, 2-O-isopropylidene-4,6-O-ethylidene-α-D-galactopyranose for producing 1 → 3-rhamnosylgalactose, linkage.
Similar content being viewed by others
Literature cited
K. Jann and O. Westphal, The Antigens, Vol. 3, Academic Press, N. Y.-San Francisco-London (1975), p. 1.
N. K. Kochetkov, B. A. Dmitriev, N. N. Malysheva, A. Ya. Chernyak, E. M. Klimov, N. E. Bayramova, and V. I. Torgov, Carbohyd. Res.,45, 283 (1975).
N. K. Kochetkov, B. A. Dmitriev, O. S. Chizhov, E. M. Klimov, N. N. Malysheva, V. I. Torgov, A. Ya. Chernyak, and N. E. Bairamova, Izv. Akad. Nauk SSSR, Ser. Khim., 1386 (1974).
N. K. Kochetkov, B. A. Dmitriev, A. Ya. Chernyak, and N. E. Bairamova, Izv. Akad. Nauk SSSR, Ser. Khim., 2331 (1974).
V. I. Torgov and A. Ya. Chernyak, Izv. Akad. Nauk SSSR, Ser. Khim., 455 (1975).
H. M. Flowers, Carbohyd. Res.,4, 312 (1967).
D. H. Ball and J. K. N. Jones, J. Chem. Soc., 905 (1958).
D. H. Ball, J. Org. Chem.,31, 220 (1966).
H. M. Flowers, Carbohyd. Res.,18, 211 (1971).
H. Bjorndal, B. Lindberg, and S. Svensson, Carbohyd. Res.,5, 433 (1967).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 7, pp. 1609–1613, July, 1977.
Rights and permissions
About this article
Cite this article
Kochetkov, N.K., Dmitriev, B.A., Nikolaev, A.V. et al. Synthesis of bacterial antigenic polysaccharides and their fragments V. Use of 1,2-o-isopropylidene-4,6-O-ethylidene-α-D-galactopyranose for the synthesis of oligosaccharides with 3-o-substituted galactopyranose residue. Russ Chem Bull 26, 1480–1483 (1977). https://doi.org/10.1007/BF00928533
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00928533