Abstract
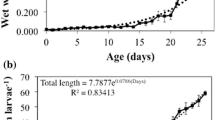
The activity of pyruvate kinase was studied in extracts of the pseudophyllidean tapewormB. acheilognathi. Extracts were prepared from immature proglottides in the anterior region and mature proglottides in the posterior regions of the tapeworms. The optiomal pH was found to be 7.5 for both extracts. Protein contents were significantly lower, and specific activities of enzyme significantly higher, in extracts prepared from anterior regions. The apparent Michaelis constant (Km a) for phosphoenol pyruvate was measured in both extracts at temperatures ranging between 10°–40°C. Values were lower, at all temperatures, for anterior extracts. The differences were greatest between 15°–20°C and 35°–40°C. The evidence suggests that at least two forms of pyruvate kinase occur inBothriocephalus.
Similar content being viewed by others
Abbreviations
- ADP:
-
adenosine diphosphate
- FDP:
-
fructose-1,6-bisphosphate
- PEP:
-
phosphoenol pyruvate
- PEPCK:
-
phosphoenol pyruvate carboxykinase
- PK:
-
pyruvate kinase
- NADH:
-
micotinamide adenosine dinucleotide (reduced)
References
Barrett J (1981) Biochemistry of parasitic helminths. Macmillan, London
Barrett J, Fairbairn D (1971) Effects of temperature on the kinetics of malate dehydrogenases in the developing eggs and adult muscle ofAscaris lumbricoides (Nematoda). J Exp Zool 176:169–177
Behm CA, Bryant C (1980) Regulatory properties of a partially purified preparation of pyruvate kinase fromFasciola hepatic. Int J Parasitol 10:107–114
Behrisch HW, Percy JA (1974) Temperature and the regulation of enzyme activity in homeo and heterothermic tissue of arctic marine mammals: some regulatory properties of 6-phosphogluconate dehydrogenase from adipose tissue of the spotted sealPhoca vitulina. Comp Biochem Physiol 47:437–443
Brazier JB, Jaffe JJ (1973) Two types of pyruvate kinase in schistosomes and filariae. Comp Biochem Physiol 44:145–155
Bryant C (1972) Metabolic regulation inMoniezia expansa (Cestoda): the role of pyruvate kinase. Int J Parasitol 2:333–340
Carter C, Fairbairn D (1975) Multienzymic nature of pyruvate kinase during development ofHymenolepis diminuta (Cestoda). J Exp Zool 194:439–448
Hall ER, Cottam LG (1978) Isozymes of pyruvate kinase in vertebrates: their physical, chemical, kinetic and immunological properties. Int J Biochem 9:785–793
Hochachka PW, Somero GN (1973) Strategies of biochemical adaptation. Saunders, Philadelphia
Körting W, Barrett J (1977) Carbohydrate catabolism in the plerocercoids ofSchistocephalus solidus (Cestoda: Pseudophyllidea). Int J Parasitol 7:511–417
Logan J, Ubelaker JE, Vrijenhoek RC (1977) Isozymes of L(+) LDH inHymenolepis diminuta. Comp Biochem Physiol 57:51–53
Low PS, Somero GN (1976) Adaptation of muscle pyruvate kinases to environmental temperatures and pressures. J Exp Zool 198:1–12
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
McManus DP (1975) Tricarboxylic acid cycle enzyme in the plerocercoid ofLigula intestinalis (Cestoda: Pseudophyllidea). Z Parasitenkd 45:319–322
Moon TW, Mustafa T, Hulbert WC, Podesta RB, Metrick DF (1977) The phosphoenol-pyruvate branch in adultHymenolepis diminuta (Cestoda): A study of pyruvate kinase and phosphoenol-pyruvate carboxykinase. J Exp Zool 200:325–336
Smyth JD (1976) Introduction to animal parasitology. 2nd edn. Hodder and Stoughton, London
Walkey M, Fairbairn D (1973) L (+)-Lactate dehydrogenases fromHymenolepis diminuta (Cestoda). J Exp Zool 183:365–374
Ward CW, Castro GA, Fairbairn D (1969) Carbon dioxide fixation and phosphoenol pyruvate metabolism inTrichinella spiralis larvae. J Parasitol 55:67–71
Wilkinson GN (1961) Statistical estimations in enzyme kinetics. Biochem J 80:324–332
Wilson FR, Somero GN, Prosser CL (1974) Temperature-metabolism relations of two species ofSebastes from different thermal environments. Comp Biochem Physiol 47:485–491
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Walkey, M., Körting, W. Thermal alterations of pyruvate kinases in the fish tapewormBothriocephalus acheilognathi, Yamaguti, 1934. Z. Parasitenkd. 71, 527–532 (1985). https://doi.org/10.1007/BF00928355
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00928355