Abstract
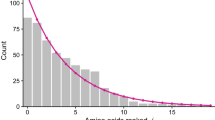
A method has been developed to determine the actin evolution rate. The method is based on amino acid composition. The actin evolution rate has been established to be extremely low. Only three or less amino acid changes per hundred amino acid residues have accumulated for a 100 million years. One can explain the conservative nature of actin evolution as a sequence of its unique tightly fitted structure rich in biologically active centres at short distances from each other. The peculiar invariability of polar amino acids leads to a conclusion that some given distribution of charges is necessary for the unique functioning of actin molecules.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adelstein, R. S., and Kuhel, W. M.: 1970,Biochemistry 9, 1355–1364.
Asatoor, A. M. and Armstrong, M. D.: 1947,Biochem. Biophys. Commun. 26, 168–175.
Atassi, M. Z.: 1975,Immunochemistry 12, 1–6.
Bailey, P.: 1970,Mathematics in Biology and Medicine, MIR, Moscow.
Bendall, J. R.: 1970,Muscles, Molecules and Movement, MIR, Moscow.
Carsten, M. E. and Katz, A. M.: 1964,Biochem. Biophys. Acta,90, 534–546.
Crumpton, M. J.: 1975,The Antigens (ed. by M. Sela), Acad. Press, N.Y., Vol.2 1–75.
Dayhoff, M.: 1972,Atlas of Protein Sequence and Structure, Nat. Biomed. Foundation, Silver Spring.
Dickerson, R. E. and Timkovich, R.: 1975,The Enzymes (ed. by P. Boyer), third ed., V. XI, pp. 397–547, Acad. Press, N.Y.
Edman, P. and Begg, G.: 1967,Eur. J. Biochem. 1, 80–93.
Elzinga, M. E., Collins, J. H., Kuhel, W. M., and Adelstein, R. S.: 1973,Proc. Nat. Acad. Sci. U.S.A. 70, 2687–2691.
Johnson, P. and Perry, S. V.: 1968,Biochem. J. 110, 207.
Katz, A. M. and Carsten, M. E.: 1963,Circulation Res. 13, 474–480
Kominz, D. R., Hough, A., Symonds, R., and Laki, K.: 1954,Arch. Biochem. Biophys. 50, 148–170.
Krans, H. M. J., Van Eijk, H. G., and Westenbrink, H. G. K.: 1965,Biochem. Biophys. Acta 100, 193–200.
Kubo, R. T., Zimmerman, B., and Grey, H. M.: 1973,The Antigens (ed. by M. Sela), Acad. Press, N.Y., Vol. 1, pp. 417–474.
Laki, K., Maruyna, K., and Komins, D. R.: 1962,Arch. Biochem. Biophys. 98, 323–330.
Laursen, R. A.: 1971,Eur. J. Radiochem. 20, 89–93.
Ogievetskaya, M. M.: 1976, inProc. of the Symp. on Biophysica and Biochemistry of Muscles, Tbilisi 1974, Nauka, p. 129.
Pinaev, G. P. and Khaitlina, S. Yu.: 1972,J. Evolution of Biochem. Physiol. VIII, 369–373.
Prager, E. M., Arnheim, N., Mross, G., and Wilson, A. C.: 1972,J. Biol. Chem. 247, 2905–2910.
Shapiro, H. M.: 1971,Biochem. Biophys. Acta 236, 725–738.
Trayer, I. P. and Perry, S. V.: 1966,Biochem. Z. 345, 87–90.
Weltman, J. K. and Dowben, R. M.: 1973,Proc. Nat. Ac. Sci. U.S.A. 70, 3230–3234.
Zagalsky, P. F.: 1972,Comp. Bioch. 41B, 385–395.
Zagalsky, P. F. and Herring, P. J.: 1972,Comp. Biochem. Physiol. 41B, 397–415.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ogievetskaya, M.M. Method for the determination of protein evolution rates by amino acid composition. Evolution rate of actins. Origins Life Evol Biosphere 8, 145–154 (1977). https://doi.org/10.1007/BF00927979
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00927979