Abstract
A fraction (FAd) capable of inhibiting specific agglutination reactions of anti-epimastigote sera (anti-LE) was obtained by extracting the sediment of lyophilized epimastigote lysates (LE) with 0.05 M phosphate buffered saline, at 37° C for 1 h. These conditions favored the action of parasite proteinase whose presence was detected by tandemcrossed immunoelectrophoresis experiments. As expected from the proteinase properties, the addition of 2-mercaptoethanol or sodium iodoacetate to the extracting solution resulted, respectively, in either increased or decreased amounts of protein in the resulting FAd.
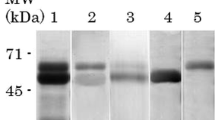
FAd components could be precipitated by the addition of Concanavalin A, methylated albumins or 0.1 N HCl. This fraction presented a single component when subjected to electrophoresis in 1% agarose gel with an electrophoretic mobility 1.2 times higher than that of human albumin. FAd component(s) were unable to penetrate 15% polycrylamide gel matrix unless 1% SDS was used. Under this condition four glycopeptide components, with Rm of 0.5, 0.55, 0.6 and 0.86, were detected.
The antigenic determinants present in FAd resisted heating at 100° C for 30 min and the prolonged action of pronase. However, these determinants were completely destroyed by the action of 25 mM sodium periodate, thus suggesting polysaccharide characteristics.
Immunization of rabbits with FAd induced the production of antibodies that were unable to precipitate with either FAd or with parasite proteinase. These antibodies exhibited positive agglutination reactions with epimastigote forms and positive immunofluorescence and immunoperoxidase reactions with trypomastigote and amastigote forms of the different strains tested. FAd was able to inhibit these reactions as well as those obtained with anti-LE and anti-FA immune sera, whereas purified proteinase was unable to inhibit any of these reactions.
Similar content being viewed by others
References
Alves MJM, Colli W (1975) Glycoproteins fromTrypanosoma cruzi. Partial purification by gel chromatography. FEBS Lett 52:188–190
Corsini AC, Costa AG, Oliveira OLP, Camargo IJB, Rangel HA (1980) A fraction (FAd) fromTrypanosoma cruzi epimastigotes depresses the immune response in mice. Immunology 40:505–554
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for the determination of sugar and related substances. Anal Chem 28:350–356
Gottlieb M (1977) A carbohydrate-containing antigen fromTrypanosoma cruzi and its detection in the circulation of infected mice. J Immunol 119:465–470
Ketteridge DS (1978) Lipopolysaccharide fromTrypanosoma cruzi. Trans R Soc Trop Med Hyg 72:101–102
Krøll J (1973) Tandem-crossed immunoelectrophoresis. In: Axelsen NH, Krøll J, Week B (eds), A manual of quantitative immunoelectrophoresis. Universitetsforlaget Olso-Bergen Tromsø, pp 57–59
Lederkremer RM, Alves MJM, Fonseca GC, Colli W (1976) A lipopeptidophosphoglycan fromTrypanosoma cruzi (epimastigote). Isolation, purification and carbohydrate composition. Biochem Biophys Acta 444:85–96
Lowry OH, Rosenbrough NJ, Farr AL, Randel RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 19:265–275
Plescia OJ (1967) Preparation of antigens for eliciting antibody with nucleoside and nucleic acid specificity. Methods Immunol Immunochem 3:175–177
Rangel HA, Araujo PMF, Camargo IJB, Bonfito M, Repka D, Sakurada JK, Atta AM (1981a) Detection of a proteinase common to epimastigote, trypomastigote and amastigote of different strains ofTrypanosoma cruzi. Tropenmed Parasitol 32:87–92
Rangel HA, Araujo PM, Repka D, Costa MG (1981b)Trypanosoma cruzi: Isolation and characterization of a proteinase. Exp Parasitol 52:199–209
Repka D, Rangel HA, Costa MG, Bellucci SBB (1979) Immunochemical study of the lysates of the epimastigote forms ofTrypanosoma cruzi. Rev Bras Biol 721:733
Repka D, Camargo IJB, Santana EM, Cunha WM, Souza OC, Sakurada JK, Rangel HA (1980) Surface antigenic determinant of epimastigote forms common to trypomastigote and amastigote forms of different strains ofTrypanosoma cruzi. Tropenmed Parasitol 239–246
Scott MT, Snary D (1979) Protective immunization of mice using cell surface glycoprotein fromTrypanosoma cruzi. Nature 282:73–74
Snary D (1980)Trypanosoma cruzi: antigenic invariance of the cell surface glycoprotein. Exp Parasitol 49:68–77
Souza W, Arguello C, Martiny-Palomo A, Trissl D, Gonzáles-Robles A, Chiari E (1977) Surface charge ofTrypanosoma cruzi. Binding of cationized ferritin and measurement of cellular electrophoretic mobility. J Protozool 24:411–415
Vickerman K (1974) Antigenic variation in African trypanosomes. In: Ciba Foundation symposium 25 (new series), Parasites in the immunized host: mechanisms of survival Associated Scientific Publishers Amsterdam, p 53–80
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cunha Tamashiro, W.M.S., Repka, D., Sakurada, J.K. et al. Trypanosoma cruzi: Surface antigenic determinants. Z. Parasitenkd. 69, 425–434 (1983). https://doi.org/10.1007/BF00927698
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00927698