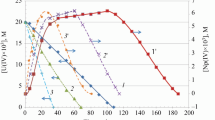
Conclusions
-
1.
The authors have used spectrophotometry to study the kinetics of the reduction of thallium(III) by sodium hypophosphite in the presence of nitrilotriacetic acid, ethylenediaminetetraacetic acid, and diethylenetriamine pentaacetic acid.
-
2.
Addition of complexones to a solution of thallium(III) makes it more resistant to reduction by H2PO2 − and increases the activation energy of the reaction.
Similar content being viewed by others
Literature cited
K. S. Gupta and Y. K. Gupta, J. Chem. Soc. A, 256 (1970).
K. S. Gupta and Y. K. Gupta, Inorg. Chem.,13, 851 (1974).
V. V. Vekshin, N. I. Pechurova, and V. I. Spitsyn, Zh. Neorg. Khim.,21, 972 (1976).
A. I. Busev and V. G. Tiptsova, Zh. Anal. Khim.,13, 18 (1958).
L. I. Martynenko, Nauch. Dokl. Vyssh. Shkoly: Khim. Khim. Tekhnol., 718 (1958).
I. V. Berezin and A. A. Klesov, Practical Course in Chemical and Fermentation Kinetics [in Russian], Izd. Mosk. Univ. (1976).
A. I. Busev, V. G. Tiptsova, and T. A. Sokolova, Zh. Neorg. Khim.,5, 2749 (1960).
J. R. Van Wazer, Phosphorus and Its Compounds, Wiley-Interscience, New York (1958).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 9, pp. 1972–1976, September, 1977.
The authors thank L. I. Martynenko for helpful consultations.
Rights and permissions
About this article
Cite this article
Pechurova, N.I., Vekshin, V.V., Zhaboedova, L.A. et al. Reduction of thallium(III) by sodium hypophosphite in aqueous solution in the presence of certain complexones. Russ Chem Bull 26, 1828–1831 (1977). https://doi.org/10.1007/BF00924369
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00924369