Conclusions
-
1.
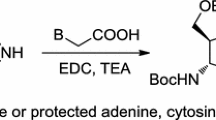
The 3′(2′)-O-amino acid esters of adenosine 5′-phosphate were synthesized employing the N-imidazolides of N-protonated amino acids.
-
2.
It was shown that the N-imidazolides of N-protonatedα-amino acids were relatively stable, and that the N-imidazolides of protonated amino acids, where the amino group is more than one carbon atom away from the carboxyl, polymerize rapidly.
-
3.
The 1,2,4-triazolides of N-protected and N-protonated amino acids do not react in aqueous medium with adenosine 5′-phosphate. The triazolides of the N-protonated amino acids do not polymerize.
Similar content being viewed by others
Literature cited
B. P. Gottikh, A. A. Krayevsky, N. B. Tarussova, P. P. Purygin, and T. L. Tsilevich, Tetrahedron,26, 4419 (1970).
B. P. Gottikh, A. A. Kraevskii, and P. P. Purygin, Izv. Akad. Nauk SSSR, Ser. Khim., 2529 (1971).
I. P. Greenstain and M. Winitz, Chemistry of the Amino Acids, Pergamon Press, New York (1961), p. 486.
Yu. I. Khurgin and I. V. Vikha, Izv. Akad. Nauk SSSR, Ser. Khim., 1245 (1968).
H. C. Beyerman and W. Maasen van den Brink, Rec. Trav. Chim.,80, 1372 (1961).
J. D. Waughan and M. O'Donnel, Tetrahedron Lett., 3727 (1968).
E. Guggenheim and J. Prue, Physicochemical Calculations [Russian translation], IL (1958), p. 429.
Author information
Authors and Affiliations
Additional information
See [1] for Communication 12.
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 8, pp. 1856–1859, August, 1974.
Rights and permissions
About this article
Cite this article
Kraevskii, A.A., Tarusova, N.B., Tsilevich, T.L. et al. Amino acid derivatives of nucleosides, nucleotides, and polynucleotides. Russ Chem Bull 23, 1776–1779 (1974). https://doi.org/10.1007/BF00923210
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00923210