Conclusions
-
1.
The corresponding 1,3-bis[ω-(p-toluenesulfonamido)alkyl]uracils are formed as a result of the reaction of 1, 3-bis(ω-bromoalkyl)uracils and the Na salt of p-toluenesulfamide.
-
2.
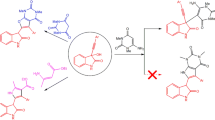
Interaction of l-(ω-bromobutyl)uracil with p-toluenesulfamide in a medium of n-butanol leads to formation of 9,18,19,20-tetraoxo-1,6,10,15-tetraazatricyclo[13.3.1.16.10]-eicosa-7, I6-diene,l-(ω-hydroxybutyl)-2-butoxypyrimid-4-one, and l-(ω-hydroxybutyl)-2-p-toluenesulfonamidopyrimid-4-one.
Similar content being viewed by others
Literature cited
J. Wempen, R. Duschinsky, L. Kaplan, and J. J. Fox, J. Amer. Chem. Soc.,83, 4755 (1961).
K. Raid, Course of Physical Organic.Chemistry [in Russian], Mir (1972), p. 415, 445.
G. Kenner and A. Todd, Heterocyclic Compounds [Russian translation], R. Elderfield (ediizor), Izd. IL, (1960), Vol. 6, p. 229.
M. Horak and J. Gut, Collect. Czech. Chem. Communs,26, 1680 (1961).
D. Shugar and J. J. Fox, Biochim. et Biophys. Acta,9, 199 (1952).
J. Pitha, J. Org. Chem.,35, 903 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 5, pp. 1103–1106, May, 1976.
Rights and permissions
About this article
Cite this article
Shvetsov, Y.S., Shirshov, A.N. & Reznik, V.S. Synthesis and properties of pyrimidinylalkylsulfonamides. Russ Chem Bull 25, 1072–1075 (1976). https://doi.org/10.1007/BF00921995
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00921995