Conclusions
-
1.
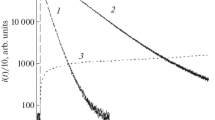
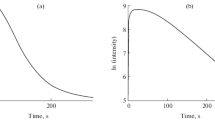
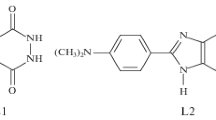
Chemiluminescence in the oxidation of luminol by hypochlorite in the presence of H2O2 does not involve free-radical reactions.
-
2.
Chemiluminescence luminol activation proceeds through interaction of the luminol with singlet excited oxygen arising from H2O2 oxidation by the hypochlorite.
Similar content being viewed by others
Literature cited
Yu. N. Kozlov, Yu. V. Koltypin, B. A. Rusin, and Yu. I. Skurlatov, Zh. Fiz. Khim.,49, 2020 (1975).
T. P. Vorob'eva, Yu. N. Kozlov, Yu. V. Koltypin, A. P. Purmal, B. A. Rusin, V. L. Tal'roze, and E. L. Frankevich, Izv. Izv. Akad. Nauk SSSR, Ser. Khim., 2187 (1976).
E. H. White, E. G. Nash, D. R. Roberts, and O. C. Zafiriou, J. Am. Chem. Soc.,90, 5932 (1968).
K. D. Gundermann, H. Fiege, and G. Klockenbring, Liebigs Ann. Chem.,738, 140 (1970).
A. A. Nadezhdin, Dissertation, Moscow (1976).
M. Kasha and A. U. Kahn, Ann. New York Acad. Sci.,171, 5 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 1, pp. 48–50, January, 1977.
Rights and permissions
About this article
Cite this article
Vorob'eva, T.P., Kozlov, Y.N., Koltypin, Y.V. et al. Processes involved in luminol oxidation accompanied by chemiluminescence. Russ Chem Bull 26, 38–40 (1977). https://doi.org/10.1007/BF00921488
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00921488