Conclusions
-
1.
A method was developed for the production of complexes of Pt(IV) with hydroxylamine by bromination of complexes of Pt(II) in CCl4.
-
2.
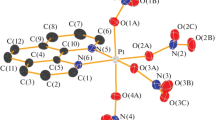
According to the data of x-electronic spectroscopy, the oxidation state of platinum in complexes of Pt(IV) is equal to +4. The presence of a Pt-Br bond in the complexes was established by the method of IR spectroscopy.
-
3.
Complexes of Pt(IV) with hydroxylamine are stable in solutions of HBr; their electronic absorption spectra were investigated under these conditions. During storage of aqueous solutions of the complexes, they are partially reduced to complexes of Pt(II).
Similar content being viewed by others
Literature cited
L. A. Chugaev and I. I. Chernyaev, Izv. In-ta po Izucheniyu Platiny Akad. Nauk SSSR, No. 1, 29 (1929).
V. I. Goremykin, Izv. In-ta po Izucheniyu Platiny Akad. Nauk SSSR, No. 14, 41 (1937).
A. V. Babaeva and N. N. Ushakova, Zh. Neorgan. Khimii,7, 487 (1962).
A. I. Stetsenko and A. A. Grinberg, Zh. Prikl. Khimii,42, 1992 (1960).
V. I. Goremykin and K. A. Gladyshevskaya, Izv. Akad. Nauk SSSR, Otd. Khim., 108 (1943).
Synthesis of Complex Compounds of Metals of the Platinum Group [in Russian], Nauka (1964).
A. I. Stetsenko, Zh. Neorgan. Khimii,6, 1769 (1961).
V. I. Nefedov, M. A. Porai-Koshits, I. A. Zakharova, I. S. Kolomnikov, and N. N. Kuz'mina, Izv. Akad. Nauk SSSR, Ser. Fiz.,36, 381 (1972).
V. I. Nefedov and M. A. Porai-Koshits, Math. Res. Bull.,7, 1543 (1972).
K. Morgan and M. Jones, J. Inorg. and Nucl. Chem.,34, 259 (1972).
R. I. Novoselov, Izv. Sibirsk. Otd. Akad. Nauk SSSR, Ser. Khim. Nauk,3, No. 7, 77 (1968).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 3, pp. 530–532, March, 1974.
Rights and permissions
About this article
Cite this article
Stetsenko, A.I., Nefedov, V.I., Abzaeva, T.G. et al. Complex compounds of platinum(IV) with hydroxylamine. Russ Chem Bull 23, 500–502 (1974). https://doi.org/10.1007/BF00921132
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00921132