Conclusions
-
1.
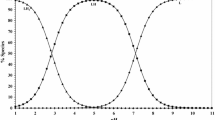
Spectrophotometric measurements have been used to determine stability constants for mono-, di-, and triacetate Pd2+ complexes at 25 and 65°C.
-
2.
The stability constant for the acetate ion complex proved to be of the same order of magnitude as that for the chloride ion complex.
Similar content being viewed by others
Literature cited
P. M. Maitlis, The Organic Chemistry of Palladium, Vol. 1, Academic Press, New York-London (1971).
É. V. Stern, Uspekhi Khimii,42, 232 (1973).
R. N. Pandey and P. M. Henry, Can. J. Chem.,52, 1241 (1974).
B. I. Habivanets, L. V. Kalabina, and L. N. Kudritskaya, Zh. Neorg. Khim.,15, 1595 (1970).
I. P. Alimarin, V. I. Shlenskaya, A. A. Biryukov, O. A. Efremenko, and V. P. Khvostova, Zh. Analit. Khim.,25, 1965 (1970).
Handbook of Chemistry [in Russian], Vol. 3, Goskhimizdat (1952).
F. Rossotti and H. Rossotti, Determination of Stability Constants and Other Equilibrium Constants in Solution [Russian translation], Mir (1965).
K. B. Yatsimirskii, Zh. Neorg. Khim.,1, 2306 (1955).
A. A. Biryukov and V. I. Shlenskaya, Zh. Neorg. Khim.,9, 813 (1964).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 7, pp. 1490–1493, July, 1976.
Rights and permissions
About this article
Cite this article
Yatsimirskii, A.K., Berezin, I.V. Spectrophotometric studies on the state of divalent palladium in aqueous acetic acid solutions. Russ Chem Bull 25, 1423–1425 (1976). https://doi.org/10.1007/BF00920810
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00920810