Abstract
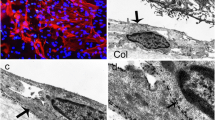
This paper addresses the molecular basis of interactions between leucocytes, other cells in the vitreoretinal environment and extracellular matrix that may underlie the pathogenesis of proliferative vitreoretinopathy. In this study we report the expression of adhesion molecules (CD11a, CD11c, CD18 and ICAM-1), lymphocyte surface markers (CD3, CD4, CD8 and CD22) and HLA-DR molecules in 25 epiretinal membranes obtained from eyes undergoing vitrectomy for the treatment of retinal detachment complicated by epiretinal membrane formation. Retinas from normal cadaveric eyes were used as controls. The results showed that cells expressing the adhesion molecules CD11a, CD11c and CD18 were present in 5 of 25, 17 of 25 and 11 of 23 membranes, respectively. Cells stained with antibodies against intracellular adhesion molecule 1 (ICAM-1) were observed in 24 of 25 membranes, whilst HLA-DR positive cells were seen in all membranes investigated. Immunohistochemical staining revealed that the molecules ICAM-1 or HLA-DR were not only expressed on inflammatory cells but also distributed within the extracellular matrix in several specimens. Lymphocytes expressing CD3 markers were present in 12 of 25 membranes, whilst T lymphocytes expressing CD4 and CD8 markers were observed in 5 of 18 and 12 of 24 membranes, respectively. In contrast, B lymphocytes expressing CD22 molecules were not found in any of the membranes. Leucocyte surface molecules were not expressed in control cadaveric retinas, although occasional cells expressing ICAM-1 were identified in the inner plexiform layer. The present findings indicate that selective interaction between infiltrating leucocytes and adhesion molecules present in extracellular matrix may well be involved in epiretinal membrane formation, and that T lymphocytes may play an important role in the pathogenesis of proliferative vitreoretinopathy.
Similar content being viewed by others
References
Arnaout MA (1990) Structure and function of the leukocyte adhesion molecules CD11/CD18. Blood 75:1037–1050
Baudouin C, Fredj-Reygrobellet D, Baudouin F, Lapalus P, Pierre Gastaud (1989) Immunohistologic study of proliferative vitreoretinopathy. Am J Ophthalmol 108:387–394
Baudouin C, Fredj-Reygrobellet D, Gordon WC, Baudouin F, Peyman G, Lapalus P, Gastaud P, Bazan N (1990) Immunohistologic study of epiretinal membranes in proliferative vitreoretinopathy. Am J Ophthalmol 110: 593–598
Broekhuyse RM, Rademakers AJJM, Van Vugt AHM, Winkens HJ (1990) Autoimmune responsiveness to retinal IRBP, S-antigen and opsin in proliferative vitreoretinopathy. Exp Eye Res 50:197–202
De Sousa M, Tilney NL, Kupiec-Weglinski JW (1991) Recognition of self within self: specific lymphocyte positioning and the extracellular matrix. Immunol Today 12:262–266
Dinarello CA, Cannon JG, Wolff SM, Bernheim HA, Bender B, Cerami A, Figari IS, Palladino MA Jr, O'Connor JV (1986) Tumour necrosis factor (cachectin) is an endogenous pyrogen and induces production of interleukin 1. J Exp Med 163:1433–1450
Docherty RJ, Forrester JV, Lackie JM (1987) Type I collagen permits invasive behaviour by retinal pigmented epithelial cells in vitro. J Cell Sci 87:399–409
Dustin ML, Rothlein R, Bhan AK, Dinarello CA, Springer TA (1986) Induction by IL-1 and interferon-γ: tissue distribution, biochemistry, and function of a natural adherence molecule (ICAM-1). J Immunol 137:245–254
Gilbert C, Hiscott P, Unger W, Grierson I, McLeod D (1988) Inflammation and the formation of epiretinal membranes. Eye [Suppl] 2:5140–156
Glaser BM, Cardin A, Biscoe B (1987) Proliferative vitreoretinopathy: the mechanism of development of vitreoretinal traction. Ophthalmology 94:327–332
Heidenkummer HP, Kampik A (1992) Intercellular adhesion molecule-1 (ICAM-1) and leukocyte function-associated antigen-1 (LFA-1) expression in human epiretinal membranes. Graefes Arch Clin Exp Ophthalmol 230:483–487
Jerdan JA, Pepose JS, Michels RG, Hayashi H, Bustros SD, Sebag M, Glaser BH (1989) Proliferative vitreoretinopathy membranes. An immunocytochemical study. Ophthalmol 96:801–810
Limb GA, Meager A, Woolley J, Wadhwa M, Biggerstaff J, Brown KA, Wolstencroft RA (1989) Release of cytokines during generation of lymphokine-activated killer (LAK) cells by IL-2. Immunol 68:514–519
Limb GA, Little BC, Meager A, Ogilvie JA, Wolstencroft RA, Franks WA, Chignell AH, Dumonde DC (1991) Cytokines in proliferative vitreoretinopathy. Eye 5:686–693
Limb GA, Hamblin AS, Wolstencroft RA, Dumonde DC (1992) Rapid cytokine up-regulation of integrins, complement receptor 1 and HLA-DR on monocytes but not on lymphocytes. Immunol 77:88–94
Liversidge JM, Sewell HF, Forrester JV (1988) Human retinal pigment epithelial cells differentially express MHC Class II (HLA DP, DR and DQ) antigens in response to in vitro stimulation with lymphokine or purified IFN-γ. Clin Exp Immunol 73:489–494
Mantovani A, Dejana E (1989) Cytokines as communication signals between leukocytes and endothelial cells. Immunol Today 10:370–375
McDonald JA (1989) Receptors for extracellular matrix components. Am J Physiol 257:L331–337
MorinoI. Hiscott P, McKechnie N, Grierson I (1990) Variation in epiretinal membrane components with clinical duration of proliferative tissue. Br J Ophthalmol 74:393–399
Papamichail M, Tsokos G, Paraskevas F (1980) Release and reconstitution of Fc receptors from normal human mononuclear cells. Immunol 40:47–52
Piela TH, Korn JH (1990) Lymphokines and cytokines in the reparative process. In: Cohen S (ed) Lymphokines and the immune response. CRC Press, Boca Raton, pp 255–273
Pober JS, Gimbrone MA, Lapierre LA, Mendrick DL, Fiers W, Rothlein R, Springer TA (1986) Overlapping patterns of activation of human endothelial cells by interleukin-1, tumour necrosis factor, and immune interferon. J Immunol 137:1893–1896
Postlethwaite AE, Lachman LB, Mainardi CL, Kang AH (1983) Interleukin 1 stimulation of collagenase production by cultured fibroblasts. J Exp Med 157:801–806
Proenca R, Travassos A, Regadas I (1990) Immunohistopathology of epiretinal membranes: evidence of major histocompatibility complex (MHC) class II antigens in proliferative vitreoretinopathies. Chibret Int J Ophthalmol 7:47–57
Retina Society Terminology Committee (1983) The classification of retinal detachment with proliferative vitreoretinopathy. Ophthalmol 90:121–125
Ryan SJ (1985) The pathophysiology of proliferative vitreoretinopathy in its management. Am J Ophthalmol 100:188–193
Scheiffarth OF, Tang S, Kampik A (1990) Macrophages and HLA-DR expression in proliferative vitreoretinopathy. Fortschr Ophthatmol 87: 340–343
Schwartz RH (1985) T-lymphocyte recognition of antigen in association with gene products of the major histocompatibility complex. Ann Rev Immunol 3:237–261
Springer TA (1990) Adhesion receptors of the immune system. Nature 346:425–434
Sugarman BJ, Aggarwal BB, Hass PE, Figari IS, Palladino MA Jr, Shepard HM (1985) Recombinant human tumor necrosis factor-α: effects on proliferation of normal and transformed cells in vitro. Science 230:943–945
Unanue ER, Allen PM (1987) The basis for the immunoregulatory role of macrophages and other accessory cells. Science 236:551–557
Wawryk SO, Novotny JR, Wicks IP, Wilkinson D, Maher D, Salvaris E, Welch K, Fecondo J, Boyd AW (1989) The role of the LFA-1/ICAM-I interaction in human leukocyte homing and adhesion. Immunol Rev 108:135–161
Wiedemann P, Weller M (1988) The pathophysiology of proliferative vitreoretinopathy. Acta Ophthalmol [Suppl] 189:7–15
Yamashita H, Hori S, Masuda K (1986) Population and proportion of component cells in preretinal membranes. Jpn J Ophthalmol 30:269–281
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Limb, G.A., Franks, W.A.A., Munasinghe, K.R. et al. Proliferative vitreoretinopathy : An examination of the involvement of lymphocytes, adhesion molecules and HLA-DR antigens. Graefe's Arch Clin Exp Ophthalmol 231, 331–336 (1993). https://doi.org/10.1007/BF00919029
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00919029