Abstract
Flow reactors are widely used in the chemical industry for purposes of catalytic reactions [1,2]. Calculation of reactors of this type, even in one-dimemional approximation, is complicated and possible only with the use of numerical methods [1, 3]. Such calculations make it possible to find the steady-state distribution of temperature and concentration in the chemical reactor if one exists; in general, however, there may be other steady-state regimes which may be preferable from the standpoint of obtaining a different degree of conversion of the starting product, operating stability, etc.
In this connection special interest attaches to the question of the existence and number of steady-state solutions of the system of equations describing the reactor process.
This problem was previously considered in [4–7]. Thus, in [4, 5] it was pointed out that in certain special cases more than one steady-state regime may exist. In [6, 7] the question of sufficient conditions of uniqueness was investigated. In [7] it was shown that the steady-state regime is unique in the ease of short reactors or a dilute mixture of reactants. In [8] the problem of the existence and uniqueness of the steady-state regime was examined for a chain reaction model with direct application of the general theorems of functional analysis.
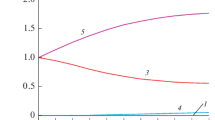
The present paper includes an analysis of a very simple mathematical model of an adiabatic chemical reactor in which an exothermic or endothermie reaction takes place. It is established that in the case of an endothermic process a unique steady-state regime always exists. In the exothermic case the problem of the steady-state regime also always has a solution which, however, may be nonunique; the possibility of the existence of several steady-state regimes, associated with the form of the temperature dependence of the heat release rate, is substantiated.
Similar content being viewed by others
References
R. Aris, Introduction to the Analysis of Chemical Reactors, New Jersey, Prentice-Hall, 1965.
I. I. Ioffe and L. M. Pis'men, Engineering Chemistry of Heterogeneous Catalysis [in Russian], izd. Khimiya, 1965.
V. S. Beskov, V. A. Kuzin, and M. G. Slin'ko, “Simulation of chemical processes in a fixed catalyst bed,” Khimischeskaya promyshlennost, no. 1, 1965.
C. Van Heerden, “The character of the stationary state of exothermic processes,” Chem. Engng Sci., vol. 8, no. 1, 1958.
T. I. Zelenyak, “On stationary solutions of mixed problems relating to the study of certain chemical processes,” Differentsial'nye uravneniya [Differential Equations], vol. 2, no. 2, 1966.
R. G. Gavalas, “On the steady states of distributed parameter systems with chemical reactions, heat and mass transfer,” Chem. Engng Sci., vol. 21, p. 477, 1966.
D. Luss and N. R. Amundson, “Uniqueness of the steady state solutions for chemical reaction occurring in a catalyst particle or in a tubular reactor with axial diffusion,” Chem. Engng Sci., vol. 22, no. 3, 1967.
L. M. Pis'men, “Stationary states of chain reactions,” PMTF [Journal of Applied Mechanics and Technical Physics], no. 3, 1966.
P. V. Danckwerts, “Continuous flow systems. Distribution of residence times,” Chem. Engng Sci., vol. 2, no. 1, 1953.
J. F. Wehner and R. H. Wilhelm, “Boundary conditions of a flow reactor,” Chem. Engng Sci., vol. 6, p. 89, 1956.
Author information
Authors and Affiliations
Additional information
The authors thank G. I. Barenblatt, A. I. Leonov, L. M. Pis'men, and Yu. I. Kharkats for discussing and commenting on the work.
Rights and permissions
About this article
Cite this article
Gupalo, Y.P., Ryazantsev, Y.S. Steady states of a continuous-flow adiabatic chemical reactor. J Appl Mech Tech Phys 8, 21–25 (1967). https://doi.org/10.1007/BF00915175
Issue Date:
DOI: https://doi.org/10.1007/BF00915175