Summary
-
1.
Data were obtained on the effect of the nature of the initiator on the course of the polymerization of styrene.
-
2.
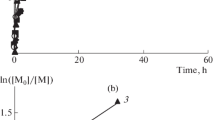
In presence of bivalent initiators the polymerization of styrene proceeds more slowly than in presence of univalent initiators, and it is accompanied by continuous rise in molecular weight through the polymerization process. The resulting polymers have higher molecular weights than those obtained with univalent radicals.
-
3.
An explanation is proposed for the observed facts.
-
4.
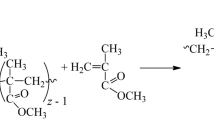
Polymeric terephthaloyl peroxide was synthesized.
Similar content being viewed by others
Literature cited
V. V. Korshak, Methods of Macromolecular Organic Chemistry, Izd. AN SSSR, 1953, p. 127–140.
H. Shah, F. Leonard and A. Tobolsci, J. Polymer. Sci. 7, 537 (1951).
I. Smets and A. Woodward, J. Polymer.Sci. 14, 126 (1954).
I. Waltcher, J. Polymer. Sci. 14, 411 (1954).
I. Breitenbach and A. Kastel, Monatsch, Chem. 84, 645 (1953).
H. Pechman and Z. Vanino, Berichte 27, 1511 (1894).
Organic Syntheses (Russian translation), 2, 547.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Korshak, V.V., Rogozhin, S.V. & Makarova, T.A. Peculiar features of the polymerization of styrene in presence of bivalent initiators. Russ Chem Bull 7, 1427–1430 (1958). https://doi.org/10.1007/BF00914857
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00914857