Abstract
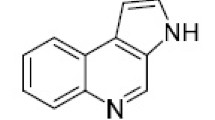
A new route to furo[2,3-b]quinolines has been developed based on the use of N-arylaconamides as starting materials. These anilides when heated with polyphosphoric acid, readily underwent intramolecular cyclization to furnish the respective 1,2-dihydro-2-oxo-quinoline-3-acetic acids which were then transformed into the corresponding dihydrofuro[2,3-b]quinolines by literature procedures. Sequential treatment of the dihydro compound withNBS andDBU afforded the corresponding furo[2,3-b]quinoline. The NMR spectra of the furoquinolines are discussed.
Similar content being viewed by others
Literatur
P. Shanmugam, T. K. Thiruvengadam undR. Palaniappan, Z. Naturforsch.28 b, 551 (1973).
P. Shanmugam, T. K. Thiruvengadam undV. T. Ramakrishnan, Synthesis1976, 393.
P. Shanmugam, Proc. Indian Acad. Sci.52, 75 (1960);
J. P. John undP. Shanmugam, l. c.54, 161 (1961).
H. Oediger, F. Moller, undK. Eiter, Synthesis1972, 591.
Y. Kuwayama, Yakugaku Zasshi81, 1501 (1961).
N. R. Campbell undJ. H. Hunt, J. Chem. Soc.1947, 1176.
P. Shanmugam, R. Palaniappan, N. Soundararajan, T. K. Thiruvengadam undK. Kanakarajan, Mh. Chem.107, 259 (1976).
A. V. Robertson, Austral. J. Chem.16, 451 (1963).
R. M. Acheson, Acridines, 2. Aufl., Vol. IX, S. 705.Wiley Interscience1973.
Characteristic of Δαβ-butenolides,R. N. Jones, G. H. Angel, I. Ito undR. J. D. Smith, Canad. J. Chem.37, 2007 (1959).
Author information
Authors and Affiliations
Additional information
Mitt.: siehe Lit.
Rights and permissions
About this article
Cite this article
Shanmugam, P., Thiruvengadam, T.K. & Ramasamy, K. Synthese von Furo [2,3-b]chinolinen. Monatshefte für Chemie 108, 725–733 (1977). https://doi.org/10.1007/BF00912817
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00912817