Abstract
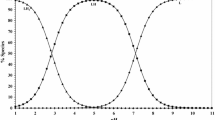
Potentiometric evidences have been cited for the formation of 1∶1∶1 ternary complexes in the systems: Cu(II)/Ni(II)-glycine-diamines [where diamines = ethylenediamine (en), propylenediamine (pn), o-phenylenediamine (phenen) and 1.8-naphthalenediamine (naphen)].
Similar content being viewed by others
Literatur
K. M. Wellman, T. G. Mecca, W. Mungall undC. R. Hare, J. Amer. Chem. Soc.89, 3646 (1967).
Sone Kozo, Utsuno, Sh. undT. Ogura, J. Inorg. Nucl. Chem.31, 117 (1969).
L. F. Chapurina undA. V. Ablov, J. Neorg. Khim.14, 1521 (1969).
M. M. Petit-Ramel undR. A. Pâris, Bull. Soc. Chim. France1969, 3070.
R.-P. Martin undR. A. Pâris, Bull. Soc. Chim. France1969, 3170.
N. E. Jackobs undD. W. Margerun, Inorg. Chem.6, 2038 (1967).
L. D. Rabindra undD. Dhrubananda, J. Indian Chem. Soc.46, 74 (1969).
G. K. Chaturvedi undJ. P. Tandon, Z. Naturforsch.25 b, 26 (1970).
G. K. Chaturvedi undJ. P. Tandon, Z. Naturforsch.23 b, 303 (1968).
A. E. Martell, S. Chaberek, Jr., R. C. Courkney, S. Westerback undH. Hyytiainen, J. Amer. Chem. Soc.79, 3036 (1957).
Author information
Authors and Affiliations
Additional information
Mit 3 Abbildungen
Rights and permissions
About this article
Cite this article
Kumari, V., Sharma, R.C. & Chaturvedi, G.K. Potentiometrische Untersuchungen an Cu(II)- und Ni(II)-Komplexen mit gemischten Liganden. Monatshefte für Chemie 105, 629–636 (1974). https://doi.org/10.1007/BF00912614
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00912614