Summary
-
1.
A thermodynamic derivation is given for the equation of molecular exchange adsorption from solutions.
-
2.
Some special forms of the equation have been found for adsorption on nonporous and porous adsorbents.
-
3.
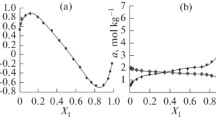
An analysis of the simpler form of the equation for the cases where the ratio of the activity coefficients in the surface and volume phases is constant or equal to unity shows that it is possible to use the molecular exchange adsorption equation to describe the forms of the isotherms from solutions encountered in practice.
Similar content being viewed by others
Literature cited
B. P. Nikol'skii and V. I. Paramonova, Uspekhi khimii8, 1535 (1939).
A. Schuchowitzky, Acta Phys. Chim. URSS8, 531 (1938).
D. Graham and R. S. Hansen, J. Phys. Chem.60, 1153 (1956).
V. É Vasserberg, A. A. Balandin, and M. P. Maksimova, Zh. fiz. khimii35, 858 (1961).
S. Yu. Elovich and O. G. Larionov, Izv. AN SSSR, Otd. khim. n. (in press).
V. K. Semenchenko, Kolloidn. zh.9, 125 (1947).
Author information
Authors and Affiliations
Additional information
Deceased.
Rights and permissions
About this article
Cite this article
Elovich, S.Y., Larionov, O.G. Theory of adsorption from nonelectrolyte solutions on solid adsorbents. Russ Chem Bull 11, 191–197 (1962). https://doi.org/10.1007/BF00908016
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00908016