Conclusions
-
1.
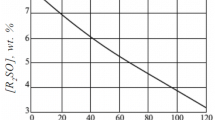
Oxidation products (alcohols, ketones, and acids) lower the rate of catalytic decomposition of n-decyl hydroperoxide under the action of copper stearate. It was hypothesized that this effect is associated either with the formation of a complex of copper stearate with oxidation products or with the influence of the oxidation products on the rate of the valence transformations of copper.
-
2.
The equilibrium constant of the formation of the complex CuSt2·2ROOH and the rate constant of the decomposition of this complex into free radicals were determined in the temperature interval 50–90°C under conditions when the oxidation products do not influence the kinetics of the decomposition of hydroperoxide.
Similar content being viewed by others
Literature cited
N. M. Émanuél', E. T. Denisov, and Z. K. Maizus, Chain Reactions of Liquid-Phase Oxidation of Hydrocarbons [in Russian], Nauka (1965).
V. M. Gol'dberg and L. K. Obukhova, Izv. Akad. Nauk SSSR, Ser. Khim., 1419 (1967); Neftekhimiya,7, 88 (1967).
G. Kamiya, Bull. Chem. Soc. Japan,38, 2156 (1965).
Z. K. Maizus and L. G. Privalova, Izv. AN SSSR. Otd. Khim. N., 628 (1963).
Z. K. Maizus, I. P. Skibida, and N. M. Émanuél', Zh. Fiz. Khimii,40, 322 (1966). 86
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 1, pp. 95–99, January, 1969.
The authors would like to express their gratitude to Z. K. Maizus for participating in the discussion of the results.
Rights and permissions
About this article
Cite this article
Bal'kov, B.G., Skibida, I.P. Influence of oxidation products on the rate of decomposition of n-decyl hydroperoxide catalyzed by copper stearate. Russ Chem Bull 18, 83–86 (1969). https://doi.org/10.1007/BF00907599
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00907599